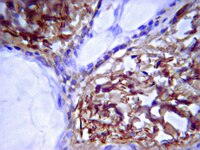
Characterization of a novel collagen chain in human placenta and its relation to AB collagen.
Sage, H and Bornstein, P
Biochemistry, 18: 3815-22 (1979)
1979
Show Abstract
A novel collagen chain, termed alpha C, has been isolated from human placenta by limited pepsin digestion. The collagen containing the alpha C chain copurifies with placental AB collagen during selective salt precipitation but is virtually absent from fetal birth membranes, which contain relatively larger amounts of AB. Both native AB and alpha C-containing collagens are resistant to human skin collagenase under conditions that support cleavage of type I by greater than 90%. The alpha C chain was separated from alpha B by phosphocellulose chromatography and subsequently from alpha P by chromatography on CM-cellulose. Its amino acid composition is distinct from alpha A and alha B although all three chains posses compositional features in common; the carbohydrate content of the alpha C chain was intermediate between those of alpha A and alpha B. Analysis by NaDodSO4-polyacrylamide gel electrophoresis of peptides produced by CNBr cleavage and by limited digestion with the enzyme mast cell protease indicated different and unique products for the alpha A, alpha B, and alpha C chains. The data support the existence of another collagen chain which is related to the alpha A and alpha B chains but which is structurally unique. The proteins containing these chains may in turn comprise a subfamily of collagen isotypes which represents a divergence from and/or specialization of the type IV basement membrane collagens. | 224919
 |
Isolation and characterization of a native placental basement-membrane collagen and its component alpha chains.
Glanville, R W, et al.
Eur. J. Biochem., 95: 383-9 (1979)
1979
Show Abstract
Native type IV collagen was isolated from human placenta using pepsin solubilisation followed by fractional salt precipitation and chromatogarphic purification. The native preparation was characterised using amino acid analyses, disc gel electrophoresis, segment-long-spacing crystallites and immunological methods. Two component alpha chains were isolated with molecular weights of approximately 95000 and 70000. Cyanogen bromide digests of these chains indicated that they are not related to any of the known alpha chains of interstitial collagens or to the recently described collagen containing alphaA and alphaB chains. They are also not related to one another and are therefore probably fragments of two genetically distinct type IV collagen alpha chains. | 456357
 |


















 Antibody[209786-ALL].jpg)