| Palmi T. Atlason, et al. (2007) N-Methyl-D-aspartate (NMDA) receptor subunit NR1 forms the substrate for oligomeric assembly of the NMDA receptor. Journal of Biological Chemistry 282, 25299-25307.
Michael A. Calderwood, et al. (2007) Epstein-Barr virus and virus human protein interaction maps. Procedings of the National Academy of Science 104, 7606-7611.
Elizabeth E. Crouch, et al. (2007) Regulation of AID expression in the immune response. Journal of Experimental Medicine 204, 1145-1156.
Amanda Nga-Sze Mak, et al. (2007) Structure-function study of maize ribosome-inactivating protein: implications for the internal inactivation region and the sole glutamate in the active site. Nucleic Acids Research 35, 6259-6267.
Preeti Malik-Kale, et al. (2007) Characterization of genetically matched isolates of Campylobacter jejuni reveals that mutations in genes involved in flagellar biosynthesis alter the organism's virulence potential. Applied and Enviornmental Microbiology 73, 3123-3136.
Dominic Nehme and Keith Poole. (2007) Assembly of the mexAB-oprM multidrug pump of Pseudomonas aeruginosa: component interactions defined by the study of pump mutant suppressors. Journal of Bacteriology 189, 6118-6127.
Satoshi Tasumi and Gerardo R. Vasta. (2007) A galectin of unique domain organization from hemocytes of the eastern oyster (Crassostrea virginica) is a receptor for the protistan parasite Perkinsus marinus. Journal of Immunology 179, 3086-3098.
J. Wu, et al. (2007) FcαRI (CD89) alleles determine the proinflammatory potential of serum IgA. Journal of Immunology 178, 3973-3982.
Hanyi Zhuang and Hiroaki Matsunami. (2007) Synergism of accessory factors in functional expression of mammalian odorant receptors. Journal of Biological Chemistry 282, 15284-15293.
Peter J. Atkinson, et al. (2006) Altered expression of Gq/11α protein shapes mGlu1 and mGlu5 receptor-mediated single cell inositol 1,4,5-trisphosphate and Ca2+ signaling. Molecular Pharmacology 69, 174-184.
R. Glenn King, Brantley R. Herrin and Louis B. Justement. (2006) Trem-Like Transcript 2 Is Expressed on Cells of the Myeloid/Granuloid and B Lymphoid Lineage and Is Up-Regulated in Response to Inflammation. Journal of Immunology 176, 6012-6021.
R. Glenn King, Brantley R. Herrin and Louis B. Justement. (2006) Trem-like transcript 2 is expressed on cells of the myeloid/granuloid and B lymphoid lineage and is up-regulated in response to inflammation. Journal of Immunology 176, 6012-6021.
James C. Samuelson, et al. (2006) Engineering a rare-cutting restriction enzyme: genetic screening and selection of NotI variants. Nucleic Acids Research 34, 796-805.
Emma L. Walker, Jeffrey L. Bose and Eric V. Stabb. (2006) Photolyase confers resistance to UV light but does not contribute to the symbiotic benefit of bioluminescence in Vibrio fischeri ES114. Applied and Enviornmental Microbiology 72, 6600-6606.
Gang Wu, et al. (2006) Simplified gene synthesis: A one-step approach to PCR-based gene construction. Journal of Biotechnology 124, 496-503.
Brantley R. Herrin, Alison L. Groeger and Louis B. Justement. (2005) The adaptor protein HSH2 attenuates apoptosis in response to ligation of the B cell antigen receptor complex on the B lymphoma cell line, WEHI-231. Journal of Biological Chemistry 280, (3507-3515).
Tom S. Kim, et al. (2005) Delayed dark adaptation in 11-cis-retinol dehydrogenase deficient mice: a role of RDH11 in visual processes in vivo. Journal of Biological Chemistry 280, 8694-8704.
Christian Siebold, et al. (2005) High-resolution structure of the catalytic region of MICAL (molecule interacting with CasL), a multidomain flavoenzyme-signaling molecule. Proceedings of the National Academy of Sciences (USA) 102, 16836-16841.
Shinya Kamauchi, et al. (2002) Structurally and Functionally Conserved Domains in the Diverse Hydrophilic Carboxy-Terminal Halves of Various Yeast and Fungal Na+/H+ Antiporters (Nha1p). 131, 821-831.
Hitoshi Niwa, et al. (2002) Phenotypic complementation establishes requirements for specific POU domain and generic transactivation function of Oct-3/4 in embryonic stem cells. Molecular and Cellular Biology 22, 1526-1536.
Yasushi Ohki, et al. (2002) Role of a conserved J8/7 X P4 base-triple in the Tetrahymena ribozyme. 132, 713-718.
Kazuaki Okuno, et al. (2002) An analysis of target preferences of Escherichia coli outer-membrane endoprotease OmpT for use in therapeutic peptide production: efficient cleavage of substrates with basic amino acids at the P4 and P6 positions. Biotechnology and Applied Biochemistry 36, 77-84.
Kazuhiro Karaya, Tatsuhito Shimizu and Akira Taketo. (2001) New gene cluster for lantibiotic streptin possibly involved in streptolysin S formation. 129, 769-775.
Shigeyuki Kawai, et al. (2001) Molecular characterization of Escherichia coli NAD kinase. European Journal of Biochemistry 268, 4359-4365.
Fumitaka Momose, et al. (2001) Cellular splicing factor RAF-2p48/NPI-5/BAT1/UAP56 interacts with the influenza virus nucleoprotein and enhances viral RNA synthesis. Journal of Virology 75, 1899-1908.
Kazufumi Yazaki, et al. (2001) A novel Coptis japonica multidrug-resistant protein preferentially expressed in the alkaloid-accumulating rhizome. Journal of Experimental Botany 52, 877-879.
Akihiro Matsuura, et al. (2000) NKT cells in the rat: organ-specific distribution of NK T cells expressing distinct Vα14 chains. Journal of Immunology 164, 3140-3148.
Kimihiko Hattori, et al. (1999) Molecular dissection of the interactions among IκBα, FWD1, and Skp1 required for ubiquitin-mediated proteolysis of IκBα. Journal of Biological Chemistry 274, 29641-29647.
Masatoshi Kitagawa, et al. (1999) An F-box protein, FWD1, mediates ubiquitin-dependent proteolysis of β-catenin. European Journal of Biochemistry 18, 2401-2410.
Nobutaka Nakashimaa, Eishi Noguchia and Takeharu Nishimotoa. (1999) Saccharomyces cerevisiae putative G protein, Gtr1p, which forms complexes with itself and a novel protein designated as Gtr2p, negatively regulates the Ran/Gsp1p G protein cycle through Gtr2p. Genetics 152, 853-867.
Kazuya Hara, et al. (1998) Archetype JC virus efficiently replicates in COS-7 cells, simian cells constitutively expressing simian virus 40 T antigen. Journal of Virology 72, 5335-5342.
Kaoru Kubokawa, Tomoyuki Miyashita and Yoshihiro Kubo. (1998) Isolation of a cDNA for a novel 120-kDa GTP-binding protein expressed in motor neurons in the salmon brain. Federation of European Biochemical Societies Letters 431, 231-235.
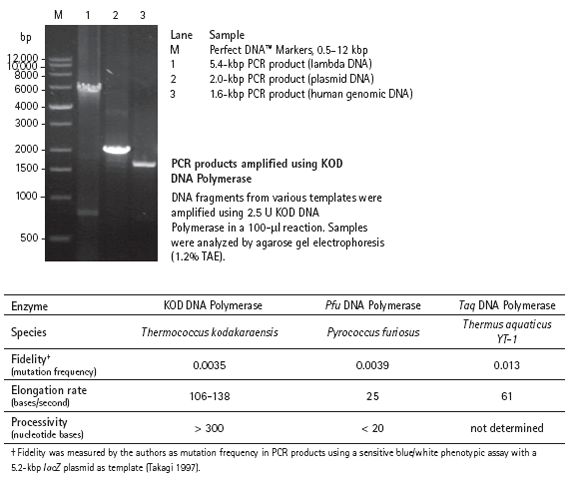
|
|