Activating RNAs associate with Mediator to enhance chromatin architecture and transcription.
Lai, F; Orom, UA; Cesaroni, M; Beringer, M; Taatjes, DJ; Blobel, GA; Shiekhattar, R
Nature
494
497-501
2013
Show Abstract
Recent advances in genomic research have revealed the existence of a large number of transcripts devoid of protein-coding potential in multiple organisms. Although the functional role for long non-coding RNAs (lncRNAs) has been best defined in epigenetic phenomena such as X-chromosome inactivation and imprinting, different classes of lncRNAs may have varied biological functions. We and others have identified a class of lncRNAs, termed ncRNA-activating (ncRNA-a), that function to activate their neighbouring genes using a cis-mediated mechanism. To define the precise mode by which such enhancer-like RNAs function, we depleted factors with known roles in transcriptional activation and assessed their role in RNA-dependent activation. Here we report that depletion of the components of the co-activator complex, Mediator, specifically and potently diminished the ncRNA-induced activation of transcription in a heterologous reporter assay using human HEK293 cells. In vivo, Mediator is recruited to ncRNA-a target genes and regulates their expression. We show that ncRNA-a interact with Mediator to regulate its chromatin localization and kinase activity towards histone H3 serine 10. The Mediator complex harbouring disease- displays diminished ability to associate with activating ncRNAs. Chromosome conformation capture confirmed the presence of DNA looping between the ncRNA-a loci and its targets. Importantly, depletion of Mediator subunits or ncRNA-a reduced the chromatin looping between the two loci. Our results identify the human Mediator complex as the transducer of activating ncRNAs and highlight the importance of Mediator and activating ncRNA association in human disease. | | 23417068
 |
Light-activation of the Archaerhodopsin H(+)-pump reverses age-dependent loss of vertebrate regeneration: sparking system-level controls in vivo.
Adams, DS; Tseng, AS; Levin, M
Biology open
2
306-13
2013
Show Abstract
Optogenetics, the regulation of proteins by light, has revolutionized the study of excitable cells, and generated strong interest in the therapeutic potential of this technology for regulating action potentials in neural and muscle cells. However, it is currently unknown whether light-activated channels and pumps will allow control of resting potential in embryonic or regenerating cells in vivo. Abnormalities in ion currents of non-excitable cells are known to play key roles in the etiology of birth defects and cancer. Moreover, changes in transmembrane resting potential initiate Xenopus tadpole tail regeneration, including regrowth of a functioning spinal cord, in tails that have been inhibited by natural inactivity of the endogenous H(+)-V-ATPase pump. However, existing pharmacological and genetic methods allow neither non-invasive control of bioelectric parameters in vivo nor the ability to abrogate signaling at defined time points. Here, we show that light activation of a H(+)-pump can prevent developmental defects and induce regeneration by hyperpolarizing transmembrane potentials. Specifically, light-dependent, Archaerhodopsin-based, H(+)-flux hyperpolarized cells in vivo and thus rescued Xenopus embryos from the craniofacial and patterning abnormalities caused by molecular blockade of endogenous H(+)-flux. Furthermore, light stimulation of Arch for only 2 days after amputation restored regenerative capacity to inhibited tails, inducing cell proliferation, tissue innervation, and upregulation of notch1 and msx1, essential genes in two well-known endogenous regenerative pathways. Electroneutral pH change, induced by expression of the sodium proton exchanger, NHE3, did not rescue regeneration, implicating the hyperpolarizing activity of Archaerhodopsin as the causal factor. The data reveal that hyperpolarization is required only during the first 48 hours post-injury, and that expression in the spinal cord is not necessary for the effect to occur. Our study shows that complex, coordinated sets of stable bioelectric events that alter body patterning-prevention of birth defects and induction of regeneration-can be elicited by the temporal modulation of a single ion current. Furthermore, as optogenetic reagents can be used to achieve that manipulation, the potential for this technology to impact clinical approaches for preventive, therapeutic, and regenerative medicine is extraordinary. We expect this first critical step will lead to an unprecedented expansion of optogenetics in biomedical research and in the probing of novel and fundamental biophysical determinants of growth and form. | | 23519324
 |
Biochemical characterization of protein quality control mechanisms during disease progression in the C22 mouse model of CMT1A.
Chittoor, VG; Sooyeon, L; Rangaraju, S; Nicks, JR; Schmidt, JT; Madorsky, I; Narvaez, DC; Notterpek, L
ASN neuro
5
e00128
2013
Show Abstract
Charcot-Marie-Tooth disease type 1A (CMT1A) is a hereditary demyelinating neuropathy linked with duplication of the peripheral myelin protein 22 (PMP22) gene. Transgenic C22 mice, a model of CMT1A, display many features of the human disease, including slowed nerve conduction velocity and demyelination of peripheral nerves. How overproduction of PMP22 leads to compromised myelin and axonal pathology is not fully understood, but likely involves subcellular alterations in protein homoeostatic mechanisms within affected Schwann cells. The subcellular response to abnormally localized PMP22 includes the recruitment of the ubiquitin-proteasome system (UPS), autophagosomes and heat-shock proteins (HSPs). Here we assessed biochemical markers of these protein homoeostatic pathways in nerves from PMP22-overexpressing neuropathic mice between the ages of 2 and 12 months to ascertain their potential contribution to disease progression. In nerves of 3-week-old mice, using endoglycosidases and Western blotting, we found altered processing of the exogenous human PMP22, an abnormality that becomes more prevalent with age. Along with the ongoing accrual of misfolded PMP22, the activity of the proteasome becomes compromised and proteins required for autophagy induction and lysosome biogenesis are up-regulated. Moreover, cytosolic chaperones are consistently elevated in nerves from neuropathic mice, with the most prominent change in HSP70. The gradual alterations in protein homoeostatic response are accompanied by Schwann cell de-differentiation and macrophage infiltration. Together, these results show that while subcellular protein quality control mechanisms respond appropriately to the presence of the overproduced PMP22, with aging they are unable to prevent the accrual of misfolded proteins. | | 24175617
 |
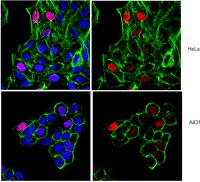
Nuclear shape changes are induced by knockdown of the SWI/SNF ATPase BRG1 and are independent of cytoskeletal connections.
Imbalzano, KM; Cohet, N; Wu, Q; Underwood, JM; Imbalzano, AN; Nickerson, JA
PloS one
8
e55628
2013
Show Abstract
Changes in nuclear morphology occur during normal development and have been observed during the progression of several diseases. The shape of a nucleus is governed by the balance of forces exerted by nuclear-cytoskeletal contacts and internal forces created by the structure of the chromatin and nuclear envelope. However, factors that regulate the balance of these forces and determine nuclear shape are poorly understood. The SWI/SNF chromatin remodeling enzyme ATPase, BRG1, has been shown to contribute to the regulation of overall cell size and shape. Here we document that immortalized mammary epithelial cells show BRG1-dependent nuclear shape changes. Specifically, knockdown of BRG1 induced grooves in the nuclear periphery that could be documented by cytological and ultrastructural methods. To test the hypothesis that the observed changes in nuclear morphology resulted from altered tension exerted by the cytoskeleton, we disrupted the major cytoskeletal networks and quantified the frequency of BRG1-dependent changes in nuclear morphology. The results demonstrated that disruption of cytoskeletal networks did not change the frequency of BRG1-induced nuclear shape changes. These findings suggest that BRG1 mediates control of nuclear shape by internal nuclear mechanisms that likely control chromatin dynamics. | Immunofluorescence | 23405182
 |
Transmembrane potential of GlyCl-expressing instructor cells induces a neoplastic-like conversion of melanocytes via a serotonergic pathway.
Blackiston, D; Adams, DS; Lemire, JM; Lobikin, M; Levin, M
Disease models & mechanisms
4
67-85
2011
Show Abstract
Understanding the mechanisms that coordinate stem cell behavior within the host is a high priority for developmental biology, regenerative medicine and oncology. Endogenous ion currents and voltage gradients function alongside biochemical cues during pattern formation and tumor suppression, but it is not known whether bioelectrical signals are involved in the control of stem cell progeny in vivo. We studied Xenopus laevis neural crest, an embryonic stem cell population that gives rise to many cell types, including melanocytes, and contributes to the morphogenesis of the face, heart and other complex structures. To investigate how depolarization of transmembrane potential of cells in the neural crest's environment influences its function in vivo, we manipulated the activity of the native glycine receptor chloride channel (GlyCl). Molecular-genetic depolarization of a sparse, widely distributed set of GlyCl-expressing cells non-cell-autonomously induces a neoplastic-like phenotype in melanocytes: they overproliferate, acquire an arborized cell shape and migrate inappropriately, colonizing numerous tissues in a metalloprotease-dependent fashion. A similar effect was observed in human melanocytes in culture. Depolarization of GlyCl-expressing cells induces these drastic changes in melanocyte behavior via a serotonin-transporter-dependent increase of extracellular serotonin (5-HT). These data reveal GlyCl as a molecular marker of a sparse and heretofore unknown cell population with the ability to specifically instruct neural crest derivatives, suggest transmembrane potential as a tractable signaling modality by which somatic cells can control stem cell behavior at considerable distance, identify a new biophysical aspect of the environment that confers a neoplastic-like phenotype upon stem cell progeny, reveal a pre-neural role for serotonin and its transporter, and suggest a novel strategy for manipulating stem cell behavior. | Immunohistochemistry | 20959630
 |
EAPP: gatekeeper at the crossroad of apoptosis and p21-mediated cell-cycle arrest.
Andorfer, P; Rotheneder, H
Oncogene
30
2679-90
2011
Show Abstract
We previously identified and characterized E2F-associated phospho-protein (EAPP), a nuclear phosphoprotein that interacts with the activating members of the E2F transcription factor family. EAPP levels are frequently elevated in transformed human cells. To examine the biological relevance of EAPP, we studied its properties in stressed and unstressed cells. Overexpression of EAPP in U2OS cells increased the fraction of G1 cells and lead to heightened resistance against DNA damage- or E2F1-induced apoptosis in a p21-dependent manner. EAPP itself becomes upregulated in confluent cells and after DNA damage and stimulates the expression of p21 independently of p53. It binds to the p21 promoter and seems to be required for the assembly of the transcription initiation complex. RNAi-mediated knockdown of EAPP expression brought about increased sensitivity towards DNA damage and resulted in apoptosis even in the absence of stress. Our results indicate that the level of EAPP is critical for cellular homeostasis. Too much of it results in G1 arrest and resistance to apoptosis, which, paradoxically, might favor cellular transformation. Too little EAPP seems to retard the expression not only of the p21 gene, but also of a number of other genes and ultimately results in apoptosis. | | 21258403
 |
The metazoan ATAC and SAGA coactivator HAT complexes regulate different sets of inducible target genes.
Zita Nagy,Anne Riss,Sally Fujiyama,Arnaud Krebs,Meritxell Orpinell,Pascal Jansen,Adrian Cohen,Henk G Stunnenberg,Shigeaki Kato,Làszlò Tora
Cellular and molecular life sciences : CMLS
67
2010
Show Abstract
Histone acetyl transferases (HATs) play a crucial role in eukaryotes by regulating chromatin architecture and locus-specific transcription. The GCN5 HAT was identified as a subunit of the SAGA (Spt-Ada-Gcn5-Acetyltransferase) multiprotein complex. Vertebrate cells express a second HAT, PCAF, that is 73% identical to GCN5. Here, we report the characterization of the mammalian ATAC (Ada-Two-A-Containing) complexes containing either GCN5 or PCAF in a mutually exclusive manner. In vitro ATAC complexes acetylate lysine 14 of histone H3. Moreover, ATAC- or SAGA-specific knock-down experiments suggest that both ATAC and SAGA are involved in the acetylation of histone H3K9 and K14 residues. Despite their catalytic similarities, SAGA and ATAC execute their coactivator functions on distinct sets of inducible target genes. Interestingly, ATAC strongly influences the global phosphorylation level of histone H3S10, suggesting that in mammalian cells a cross-talk exists linking ATAC function to H3S10 phosphorylation. | | 19936620
 |
The spatial relationship between heterochromatin protein 1 alpha and histone modifications during mouse oocyte meiosis.
Qiang Wang,Jun-Shu Ai,Safiriyu Idowu Ola,Ling Gu,Yong-Zhong Zhang,Da-Yuan Chen,Qing-Yuan Sun
Cell cycle (Georgetown, Tex.)
7
2008
Show Abstract
Heterochromatin protein 1 (HP1) is closely associated with diverse chromatin organization and function in mitosis. However, we almost know nothing about HP1 in mammalian oocyte. Here, we investigated the subcellular distribution of HP1alpha and its spatial relationship to histone modifications during mouse oocyte maturation. Dynamic migration of HP1alpha was observed in germinal vesicle with non-surrounded nucleolus (NSN) to surrounded nucleolus (SN) oocytes, which may be essential for the transition of chromatin conformation during the development of antral oocytes. In meiosis, HP1alpha was clearly detectable at the periphery of chromosomes from pre-metaphase I stage to anaphase-telophase I stage. Spatial correlation between HP1alpha and histone modifications is highly variable around the time of meiotic resumption. In germinal vesicle oocytes, HP1alpha almost colocalized with all histone modifications examined in this study except for phosphorylation of serine 28 on histone H3. However, with the breakdown of germinal vesicle, HP1alpha was detected mostly in the chromosomal domains with strong phosphorylation of serine 10 and 28 on histone H3, and they also partially associated with methylated histones. These results presented the functional implication of histone modifications in the regulation of HP1alpha during oocyte maturation. In addition, we also showed that blocking the function of HP1alpha by microinjecting anti-HP1alpha antibody caused the delay of GVBD, however, this effect may not be achieved through modifying histones. | | 18239470
 |
Drug-induced activation of dopamine D(1) receptor signaling and inhibition of class I/II histone deacetylase induce chromatin remodeling in reward circuitry and modulate cocaine-related behaviors.
Schroeder, FA; Penta, KL; Matevossian, A; Jones, SR; Konradi, C; Tapper, AR; Akbarian, S
Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology
33
2981-92
2008
Show Abstract
Chromatin remodeling, including histone modification, is involved in stimulant-induced gene expression and addiction behavior. To further explore the role of dopamine D(1) receptor signaling, we measured cocaine-related locomotor activity and place preference in mice pretreated for up to 10 days with the D(1) agonist SKF82958 and/or the histone deacetylase inhibitor (HDACi), sodium butyrate. Cotreatment with D(1) agonist and HDACi significantly enhanced cocaine-induced locomotor activity and place preference, in comparison to single-drug regimens. However, butyrate-mediated reward effects were transient and only apparent within 2 days after the last HDACi treatment. These behavioral changes were associated with histone modification changes in striatum and ventral midbrain: (1) a generalized increase in H3 phosphoacetylation in striatal neurons was dependent on activation of D(1) receptors; (2) H3 deacetylation at promoter sequences of tyrosine hydroxylase (Th) and brain-derived neurotrophic factor (Bdnf) in ventral midbrain, together with upregulation of the corresponding gene transcripts after cotreatment with D(1) agonist and HDACi. Collectively, these findings imply that D(1) receptor-regulated histone (phospho)acetylation and gene expression in reward circuitry is differentially regulated in a region-specific manner. Given that the combination of D(1) agonist and HDACi enhances cocaine-related sensitization and reward, the therapeutic benefits of D(1) receptor antagonists and histone acetyl-transferase inhibitors (HATi) warrant further investigation in experimental models of stimulant abuse. | | 18288092
 |
Histone hyperacetylation occurs on promoters of lytic cycle regulatory genes in Epstein-Barr virus-infected cell lines which are refractory to disruption of latency by histone deacetylase inhibitors.
Countryman, JK; Gradoville, L; Miller, G
Journal of virology
82
4706-19
2008
Show Abstract
Activation of the Epstein-Barr virus (EBV) lytic cycle is mediated through the combined actions of ZEBRA and Rta, the products of the viral BZLF1 and BRLF1 genes. During latency, these two genes are tightly repressed. Histone deacetylase inhibitors (HDACi) can activate viral lytic gene expression. Therefore, a widely held hypothesis is that Zp and Rp, the promoters for BZLF1 and BRLF1, are repressed by chromatin and that hyperacetylation of histone tails, by allowing the access of positively acting factors, leads to transcription of BZLF1 and BRLF1. To investigate this hypothesis, we used chromatin immunoprecipitation (ChIP) to examine the acetylation and phosphorylation states of histones H3 and H4 on Zp and Rp in three cell lines, Raji, B95-8, and HH514-16, which differ in their response to EBV lytic induction by HDACi. We studied the effects of three HDACi, sodium butyrate (NaB), trichostatin A (TSA), and valproic acid (VPA). We also examined the effects of tetradecanoyl phorbol acetate (TPA) and 5-aza-2'-deoxycytidine, a DNA methyltransferase inhibitor, on histone modification. In Raji cells, TPA and NaB act synergistically to activate the EBV lytic cycle and promote an increase in histone H3 and H4 acetylation and phosphorylation at Zp and Rp. Surprisingly, however, when Raji cells were treated with NaB or TSA, neither of which is sufficient to activate the lytic cycle, an increase of comparable magnitude of hyperacetylated and phosphorylated histone H3 at Zp and Rp was observed. In B95-8 cells, NaB inhibited lytic induction by TPA, yet NaB promoted hyperacetylation of H3 and H4. In HH514-16 cells, NaB and TSA strongly activated the EBV lytic cycle and caused hyperacetylation of histone H3 on Zp and Rp. However, when HH514-16 cells were treated with VPA, lytic cycle mRNAs or proteins were not induced, although histone H3 was hyperacetylated as measured by immunoblotting or by ChIP on Zp and Rp. Taken together, our data suggest that open chromatin at EBV BZLF1 and BRLF1 promoters is not sufficient to activate EBV lytic cycle gene expression. | | 18337569
 |