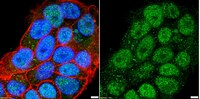
Characterization of aromatase expression in the adult male and female mouse brain. I. Coexistence with oestrogen receptors α and β, and androgen receptors.
Stanić, D; Dubois, S; Chua, HK; Tonge, B; Rinehart, N; Horne, MK; Boon, WC
PloS one
9
e90451
2014
Show Abstract
Aromatase catalyses the last step of oestrogen synthesis. There is growing evidence that local oestrogens influence many brain regions to modulate brain development and behaviour. We examined, by immunohistochemistry, the expression of aromatase in the adult male and female mouse brain, using mice in which enhanced green fluorescent protein (EGFP) is transcribed following the physiological activation of the Cyp19A1 gene. EGFP-immunoreactive processes were distributed in many brain regions, including the bed nucleus of the stria terminalis, olfactory tubercle, medial amygdaloid nucleus and medial preoptic area, with the densest distributions of EGFP-positive cell bodies in the bed nucleus and medial amygdala. Differences between male and female mice were apparent, with the density of EGFP-positive cell bodies and fibres being lower in some brain regions of female mice, including the bed nucleus and medial amygdala. EGFP-positive cell bodies in the bed nucleus, lateral septum, medial amygdala and hypothalamus co-expressed oestrogen receptor (ER) α and β, or the androgen receptor (AR), although single-labelled EGFP-positive cells were also identified. Additionally, single-labelled ERα-, ERβ- or AR-positive cell bodies often appeared to be surrounded by EGFP-immunoreactive nerve fibres/terminals. The widespread distribution of EGFP-positive cell bodies and fibres suggests that aromatase signalling is common in the mouse brain, and that locally synthesised brain oestrogens could mediate biological effects by activating pre- and post-synaptic oestrogen α and β receptors, and androgen receptors. The higher number of EGFP-positive cells in male mice may indicate that the autocrine and paracrine effects of oestrogens are more prominent in males than females. | | | 24646567
 |
Social isolation during puberty affects female sexual behavior in mice.
Kercmar, J; Tobet, SA; Majdic, G
Frontiers in behavioral neuroscience
8
337
2014
Show Abstract
Exposure to stress during puberty can lead to long-term behavioral alterations in adult rodents coincident with sex steroid hormone-dependent brain remodeling and reorganization. Social isolation is a stress for social animals like mice, but little is known about the effects of such stress during adolescence on later reproductive behaviors. The present study examined sexual behavior of ovariectomized, estradiol and progesterone primed female mice that were individually housed from 25 days of age until testing at approximately 95 days, or individually housed from day 25 until day 60 (during puberty), followed by housing in social groups. Mice in these isolated groups were compared to females that were group housed throughout the experiment. Receptive sexual behaviors of females and behaviors of stimulus males were recorded. Females housed in social groups displayed greater levels of receptive behaviors in comparison to both socially isolated groups. Namely, social females had higher lordosis quotients (LQs) and more often displayed stronger lordosis postures in comparison to isolated females. No differences between female groups were observed in stimulus male sexual behavior suggesting that female "attractiveness" was not affected by their social isolation. Females housed in social groups had fewer cells containing immunoreactive estrogen receptor (ER) α in the anteroventral periventricular nucleus (AVPV) and in the ventromedial nucleus of the hypothalamus (VMH) than both isolated groups. These results suggest that isolation during adolescence affects female sexual behavior and re-socialization for 1 month in adulthood is insufficient to rescue lordosis behavior from the effects of social isolation during the pubertal period. | Immunohistochemistry | | 25324747
 |
Estrogens stimulate serotonin neurons to inhibit binge-like eating in mice.
Cao, X; Xu, P; Oyola, MG; Xia, Y; Yan, X; Saito, K; Zou, F; Wang, C; Yang, Y; Hinton, A; Yan, C; Ding, H; Zhu, L; Yu, L; Yang, B; Feng, Y; Clegg, DJ; Khan, S; DiMarchi, R; Mani, SK; Tong, Q; Xu, Y
The Journal of clinical investigation
124
4351-62
2014
Show Abstract
Binge eating afflicts approximately 5% of US adults, though effective treatments are limited. Here, we showed that estrogen replacement substantially suppresses binge-like eating behavior in ovariectomized female mice. Estrogen-dependent inhibition of binge-like eating was blocked in female mice specifically lacking estrogen receptor-α (ERα) in serotonin (5-HT) neurons in the dorsal raphe nuclei (DRN). Administration of a recently developed glucagon-like peptide-1-estrogen (GLP-1-estrogen) conjugate designed to deliver estrogen to GLP1 receptor-enhanced regions effectively targeted bioactive estrogens to the DRN and substantially suppressed binge-like eating in ovariectomized female mice. Administration of GLP-1 alone reduced binge-like eating, but not to the same extent as the GLP-1-estrogen conjugate. Administration of ERα-selective agonist propylpyrazole triol (PPT) to murine DRN 5-HT neurons activated these neurons in an ERα-dependent manner. PPT also inhibited a small conductance Ca2+-activated K+ (SK) current; blockade of the SK current prevented PPT-induced activation of DRN 5-HT neurons. Furthermore, local inhibition of the SK current in the DRN markedly suppressed binge-like eating in female mice. Together, our data indicate that estrogens act upon ERα to inhibit the SK current in DRN 5-HT neurons, thereby activating these neurons to suppress binge-like eating behavior and suggest ERα and/or SK current in DRN 5-HT neurons as potential targets for anti-binge therapies. | | | 25157819
 |
Estradiol treatment prevents injury induced enhancement in spinal cord dynorphin expression.
Gupta, DS; Hubscher, CH
Frontiers in physiology
3
28
2011
Show Abstract
Administration of the ovarian steroid estradiol in male and female animals has been shown to have neuromodulatory and neuroprotective effects in a variety of experimental models. In the present study, spinal tissues from dermatomes just above (T5-T7, at level) a severe chronic spinal cord injury (SCI) at T8 were analyzed for expression levels of prodynorphin (PRDN) and phospho-(serine 369) κ-opioid receptor (KOR-P) in 17 β estradiol (EB)- and placebo-treated adult male rats. Dynorphin was targeted since (1) it has previously been shown to be elevated post-SCI, (2) intrathecal injection of dynorphin produces several of the same adverse effects seen with a SCI, and (3) its increased expression is known to occur in a variety of different experimental models of central neuropathic pain. A significant elevation of extracellular levels of both PRDN and KOR-P in the placebo-treated SCI group relative to uninjured surgical sham controls was found in spinal tissues above the injury level, indicating increased dynorphin levels. Importantly, the EB-treated SCI group did not show elevations of PRDN levels at 6 weeks post-injury. Immunohistochemical analysis of at level tissues revealed that EB treatment significantly prevented a post-SCI increase in expression of PRDN puncta co-labeling synapsin I, a nerve terminal marker. The dynorphin-containing terminals co-labeled vesicular glutamate receptor-2 (a marker of glutamatergic terminals), a finding consistent with a non-opioid basis for the adverse effects of dynorphin. These results support a beneficial role for EB treatment post-SCI through a reduction in excessive spinal cord levels of dynorphin. Studies manipulating the timing of the EB treatment post-injury along with specific functional assessments will address whether the beneficial effects are due to EB's potential neuromodulatory or neuroprotective action. | Immunohistochemistry | Rat | 22371702
 |
Neurochemical characterization and sexual dimorphism of projections from the brain to abdominal and subcutaneous white adipose tissue in the rat.
Adler, ES; Hollis, JH; Clarke, IJ; Grattan, DR; Oldfield, BJ
The Journal of neuroscience : the official journal of the Society for Neuroscience
32
15913-21
2011
Show Abstract
Retroperitoneal white adipose tissue (rWAT) and subcutaneous (inguinal) white adipose tissue (iWAT) are both innervated and regulated by sympathetic efferents, but the distribution and identity of the cells in the brain that regulate sympathetic outflow are poorly characterized. Our aim was to use two isogenic strains of a neurotropic virus (pseudorabies, Bartha) tagged with either green or red fluorescent reporters to identify cells in the brain that project to rWAT and/or iWAT. These viruses were injected into separate WAT depots in male and female Sprague Dawley rats. Retrogradely labeled neurons in the CNS were characterized by immunohistochemistry and PCR. For the latter, laser capture of individual virally labeled neurons was used. All virally labeled brain regions contained neurons projecting to either and both WAT depots. Neurons to abdominal fat were the most abundant in males, whereas females contained a greater proportion of neurons to subcutaneous via private lines and collateral branches. Retrogradely labeled neurons directed to WAT expressed estrogen receptor-α (ERα), and fewer neurons to subcutaneous WAT expressed ERα in males. Regardless of sex, projections from the arcuate nucleus were predominantly from pro-opiomelanocortin cells, with a notable lack of projections from agouti-related protein-expressing neurons. Within the lateral hypothalamus, neurons directed to rWAT and iWAT expressed orexin and melanin-concentrating hormone (MCH), but male rats had a predominance of MCH directed to iWAT. In conclusion, the neurochemical substrates that project through polysynaptic pathways to iWAT and rWAT are different in male and female rats, suggesting that metabolic regulation of rWAT and iWAT is sexually dimorphic. | Immunohistochemistry | | 23136429
 |
The impact of neonatal bisphenol-A exposure on sexually dimorphic hypothalamic nuclei in the female rat.
Adewale, HB; Todd, KL; Mickens, JA; Patisaul, HB
Neurotoxicology
32
38-49
2010
Show Abstract
Now under intense scrutiny, due to its endocrine disrupting properties, the potential threat the plastics component bisphenol-A (BPA) poses to human health remains unclear. Found in a multitude of polycarbonate plastics, food and beverage containers, and medical equipment, BPA is thought to bind to estrogen receptors (ERs), thereby interfering with estrogen-dependent processes. Our lab has previously shown that exposure to BPA (50mg/kg bw or 50μg/kg bw) during the neonatal critical period is associated with advancement of puberty, early reproductive senescence and ovarian malformations in female Long Evans rats. Here, using neural tissue obtained from the same animals, we explored the impact of neonatal BPA exposure on the development of sexually dimorphic hypothalamic regions critical for female reproductive physiology and behavior. Endpoints included quantification of oxytocin-immunoreactive neurons (OT-ir) in the paraventricular nucleus (PVN), serotonin (5-HT-ir) fiber density in the ventrolateral subdivision of the ventromedial nucleus (VMNvl) as well as ERα-ir neuron number in the medial preoptic area (MPOA), the VMNvl, and the arcuate nucleus (ARC). Both doses of BPA increased the number of OT-ir neurons within the PVN, but no significant effects were seen on 5-HT-ir fiber density or ERα-ir neuron number in any of the areas analyzed. In addition to hypothalamic development, we also assessed female sex behavior and body weight. No effect of BPA on sexual receptivity or proceptive behavior in females was observed. Females treated with BPA, however, weighed significantly more than control females by postnatal day 99. This effect of BPA on weight is critical because alterations in metabolism, are frequently associated with reproductive dysfunction. Collectively, the results of this and our prior study indicate that the impact of neonatal BPA exposure within the female rat hypothalamus is region specific and support the hypothesis that developmental BPA exposure may adversely affect reproductive development in females. | Immunohistochemistry | | 20696184
 |
Radiation acts on the microenvironment to affect breast carcinogenesis by distinct mechanisms that decrease cancer latency and affect tumor type.
Nguyen, DH; Oketch-Rabah, HA; Illa-Bochaca, I; Geyer, FC; Reis-Filho, JS; Mao, JH; Ravani, SA; Zavadil, J; Borowsky, AD; Jerry, DJ; Dunphy, KA; Seo, JH; Haslam, S; Medina, D; Barcellos-Hoff, MH
Cancer cell
19
640-51
2010
Show Abstract
Tissue microenvironment is an important determinant of carcinogenesis. We demonstrate that ionizing radiation, a known carcinogen, affects cancer frequency and characteristics by acting on the microenvironment. Using a mammary chimera model in which an irradiated host is transplanted with oncogenic Trp53 null epithelium, we show accelerated development of aggressive tumors whose molecular signatures were distinct from tumors arising in nonirradiated hosts. Molecular and genetic approaches show that TGFβ mediated tumor acceleration. Tumor molecular signatures implicated TGFβ, and genetically reducing TGFβ abrogated the effect on latency. Surprisingly, tumors from irradiated hosts were predominantly estrogen receptor negative. This effect was TGFβ independent and linked to mammary stem cell activity. Thus, the irradiated microenvironment affects latency and clinically relevant features of cancer through distinct and unexpected mechanisms. | Immunohistochemistry | | 21575864
 |
Estradiol inhibits ongoing autoimmune neuroinflammation and NFkappaB-dependent CCL2 expression in reactive astrocytes.
Giraud, Sébastien N, et al.
Proc. Natl. Acad. Sci. U.S.A., 107: 8416-21 (2010)
2009
Show Abstract
Astroglial reactivity associated with increased production of NFkappaB-dependent proinflammatory molecules is an important component of the pathophysiology of chronic neurological disorders such as multiple sclerosis (MS). The use of estrogens as potential anti-inflammatory and neuroprotective drugs is a matter of debate. Using mouse experimental allergic encephalomyelitis (EAE) as a model of chronic neuroinflammation, we report that implants reproducing pregnancy levels of 17beta-estradiol (E2) alleviate ongoing disease and decrease astrocytic production of CCL2, a proinflammatory chemokine that drives the local recruitment of inflammatory myeloid cells. Immunohistochemistry and confocal imaging reveal that, in spinal cord white matter EAE lesions, reactive astrocytes express estrogen receptor (ER)alpha (and to a lesser extent ERbeta) with a preferential nuclear localization, whereas other cells including infiltrated leukocytes express ERs only in their membranes or cytosol. In cultured rodent astrocytes, E2 or an ERalpha agonist, but not an ERbeta agonist, inhibits TNFalpha-induced CCL2 expression at nanomolar concentrations, and the ER antagonist ICI 182,170 blocks this effect. We show that this anti-inflammatory action is not associated with inhibition of NFkappaB nuclear translocation but rather involves direct repression of NFkappaB-dependent transcription. Chromatin immunoprecipitation assays further indicate that estrogen suppresses TNFalpha-induced NFkappaB recruitment to the CCL2 enhancer. These data uncover reactive astrocytes as an important target for nuclear ERalpha inhibitory action on chemokine expression and suggest that targeting astrocytic nuclear NFkappaB activation with estrogen receptor alpha modulators may improve therapies of chronic neurodegenerative disorders involving astroglial neuroinflammation. | | | 20404154
 |
Roles for gamma-aminobutyric acid in the development of the paraventricular nucleus of the hypothalamus.
Kristy M McClellan,Matthew S Stratton,Stuart A Tobet
The Journal of comparative neurology
518
2009
Show Abstract
The development of the hypothalamic paraventricular nucleus (PVN) involves several factors that work together to establish a cell group that regulates neuroendocrine functions and behaviors. Several molecular markers were noted within the developing PVN, including estrogen receptors (ER), neuronal nitric oxide synthase (nNOS), and brain-derived neurotrophic factor (BDNF). By contrast, immunoreactive gamma-aminobutyric acid (GABA) was found in cells and fibers surrounding the PVN. Two animal models were used to test the hypothesis that GABA works through GABA(A) and GABA(B) receptors to influence the development of the PVN. Treatment with bicuculline to decrease GABA(A) receptor signaling from embryonic day (E) 10 to E17 resulted in fewer cells containing immunoreactive (ir) ERalpha in the region of the PVN vs. control. GABA(B)R1 receptor subunit knockout mice were used to examine the PVN at P0 without GABA(B) signaling. In female but not male GABA(B)R1 subunit knockout mice, the positions of cells containing ir ERalpha shifted from medial to lateral compared with wild-type controls, whereas the total number of ir ERalpha-containing cells was unchanged. In E17 knockout mice, ir nNOS cells and fibers were spread over a greater area. There was also a significant decrease in ir BDNF in the knockout mice in a region-dependent manner. Changes in cell position and protein expression subsequent to disruption of GABA signaling may be due, in part, to changes in nNOS and BDNF signaling. Based on the current study, the PVN can be added as another site where GABA exerts morphogenetic actions in development. Full Text Article | | | 20506472
 |
Expression of aromatase, androgen and estrogen receptors in peripheral target tissues in diabetes.
Prabhu, A; Xu, Q; Manigrasso, MB; Biswas, M; Flynn, E; Iliescu, R; Lephart, ED; Maric, C
Steroids
75
779-87
2009
Show Abstract
Our previous studies have shown that diabetes in the male streptozotocin (STZ)-induced diabetic rat is characterized by a decrease in circulating testosterone and concomitant increase in estradiol levels. Interestingly, this increase in estradiol levels persists even after castration, suggesting extra-testicular origins of estradiol in diabetes. The aim of the present study was to examine whether other target organs of diabetes may be sources of estradiol. The study was performed in male Sprague-Dawley non-diabetic (ND), STZ-induced diabetic (D) and STZ-induced diabetic castrated (Dcas) rats (n=8-9/group). 14 weeks of diabetes was associated with decreased testicular (ND, 26.3+/-4.19; D, 18.4+/-1.54; Pless than 0.05), but increased renal (ND, 1.83+/-0.92; D, 7.85+/-1.38; Pless than 0.05) and ocular (D, 23.4+/-3.66; D, 87.1+/-28.1; Pless than 0.05) aromatase activity. This increase in renal (Dcas, 6.30+/-1.25) and ocular (Dcas, 62.7+/-11.9) aromatase activity persisted after castration. The diabetic kidney also had increased levels of tissue estrogen (ND, 0.31+/-0.01; D, 0.51+/-0.11; Dcas, 0.45+/-0.08) as well as estrogen receptor alpha protein expression (ND, 0.63+/-0.09; D, 1.62+/-0.28; Dcas, 1.38+/-0.20). These data suggest that in male STZ-induced diabetic rats, tissues other than the testis may become sources of estradiol. In particular, the diabetic kidney appears to produce estradiol following castration, a state that is associated with a high degree or renal injury. Overall, our data provides evidence for the extra-testicular source of estradiol that in males, through an intracrine mechanism, may contribute to the development and/or progression of end-organ damage associated with diabetes. Full Text Article | | | 20064538
 |