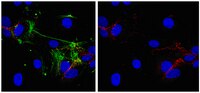
ARF6 inhibition stabilizes the vasculature and enhances survival during endotoxic shock.
Davis, CT; Zhu, W; Gibson, CC; Bowman-Kirigin, JA; Sorensen, L; Ling, J; Sun, H; Navankasattusas, S; Li, DY
Journal of immunology (Baltimore, Md. : 1950)
192
6045-52
2014
Zobrazit abstrakt
The vascular endothelium responds to infection by destabilizing endothelial cell-cell junctions to allow fluid and cells to pass into peripheral tissues, facilitating clearance of infection and tissue repair. During sepsis, endotoxin and other proinflammatory molecules induce excessive vascular leak, which can cause organ dysfunction, shock, and death. Current therapies for sepsis are limited to antibiotics and supportive care, which are often insufficient to reduce morbidity and prevent mortality. Previous attempts at blocking inflammatory cytokine responses in humans proved ineffective at reducing the pathologies associated with sepsis, highlighting the need for a new therapeutic strategy. The small GTPase ARF6 is activated by a MyD88-ARNO interaction to induce vascular leak through disruption of endothelial adherens junctions. In this study, we show that the MyD88-ARNO-ARF6-signaling axis is responsible for LPS-induced endothelial permeability and is a destabilizing convergence point used by multiple inflammatory cues. We also show that blocking ARF6 with a peptide construct of its N terminus is sufficient to reduce vascular leak and enhance survival during endotoxic shock, without inhibiting the host cytokine response. Our data highlight the therapeutic potential of blocking ARF6 and reducing vascular leak for the treatment of inflammatory conditions, such as endotoxemia. | 24835390
 |
Origin of endothelial progenitors in human postnatal bone marrow.
Reyes, M; Dudek, A; Jahagirdar, B; Koodie, L; Marker, PH; Verfaillie, CM
The Journal of clinical investigation
109
337-46
2002
Zobrazit abstrakt
This study demonstrates that a CD34(-), vascular endothelial cadherin(-) (VE-cadherin(-)), AC133(+), and fetal liver kinase(+) (Flk1(+)) multipotent adult progenitor cell (MAPC) that copurifies with mesenchymal stem cells from postnatal human bone marrow (BM) is a progenitor for angioblasts. In vitro, MAPCs cultured with VEGF differentiate into CD34(+), VE-cadherin(+), Flk1(+) cells - a phenotype that would be expected for angioblasts. They subsequently differentiate into cells that express endothelial markers, function in vitro as mature endothelial cells, and contribute to neoangiogenesis in vivo during tumor angiogenesis and wound healing. This in vitro model of preangioblast-to-endothelium differentiation should prove very useful in studying commitment to the angioblast and beyond. In vivo, MAPCs can differentiate in response to local cues into endothelial cells that contribute to neoangiogenesis in tumors. Because MAPCs can be expanded in culture without obvious senescence for more than 80 population doublings, they may be an important source of endothelial cells for cellular pro- or anti-angiogenic therapies. | 11827993
 |
CD34- blood-derived human endothelial cell progenitors.
Harraz, M; Jiao, C; Hanlon, HD; Hartley, RS; Schatteman, GC
Stem cells (Dayton, Ohio)
19
304-12
2001
Zobrazit abstrakt
A subset of adult peripheral blood leukocytes functions as endothelial cell progenitors called angioblasts. They can incorporate into the vasculature in animal models of neovascularization and accelerate the restoration of blood flow to mouse ischemic limbs. Earlier reports suggested that CD34-expressing (CD34+) but not CD34+ cell-depleted (CD34-) leukocytes can differentiate into endothelial cells (EC) in vitro and in vivo. Recent findings suggest that CD14+ cells, which are typically CD34-, also have angioblast-like properties in vitro. To determine the identity of angioblasts, the potential of CD34+, CD34-, CD34- CD14+, and CD34- CD14- cells to produce EC was compared. We show that a subset of monocyte (CD34- CD14+)-enriched cells can take on an EC-like phenotype in culture, but that the EC-like cells also express dendritic cell antigens. These findings suggest that monocytes differentiate into macrophages, dendritic cells, or EC depending on environmental cues. The data also demonstrate that angioblasts are more abundant in the blood than previously thought. Finally, we demonstrate that CD34- and CD34- CD14+ cells incorporate into the endothelium of blood vessels in mouse ischemic limbs. However, incorporation of these cells requires co-injection with CD34+ cells, indicating that leukocyte-leukocyte interactions may play a critical role in governing angioblast behavior in vivo. | 11463950
 |
Functional roles for PECAM-1 (CD31) and VE-cadherin (CD144) in tube assembly and lumen formation in three-dimensional collagen gels.
Yang, S; Graham, J; Kahn, JW; Schwartz, EA; Gerritsen, ME
The American journal of pathology
155
887-95
1998
Zobrazit abstrakt
Various in vitro models have been described that emulate one or more of the processes involved in angiogenesis in vivo. In the present study endothelial cells were cultured in three-dimensional type I collagen lattices in the presence of a mixture of basic fibroblast growth factor, vascular endothelial cell growth factor, and phorbol myristate acetate. Under these conditions, the endothelial cells rapidly assemble into an interconnected network of tube-like structures with a high frequency of intercellular canals or lumens. The formation of the networks and lumens was completely blocked by cycloheximide and by actinomycin D. Monoclonal antibodies directed against CD31 or vascular endothelial cadherin (VE-cadherin) inhibited the formation of endothelial tubes. A subtle difference in the morphology of cells treated with anti-CD31 versus anti-VE-cadherin was noted; namely, cells incubated in the presence of CD31 antibodies were rounded or formed attenuated tube-like structures, both of which were characterized by a single, large intra- or intercellular vacuole. In contrast, tube formation by cells incubated in the presence of VE-cadherin antibodies was also impaired and, most notably, demonstrated a reduction in either vacuole formation or vacuole fusion, depending upon the monoclonal antibody used. We suggest that the two endothelial-junction-associated proteins, CD31 and VE-cadherin, play different roles in the process of tube formation. CD31 appears to be required for cell elongation, migration, and/or invasion in the gels as well as for cell-cell association to form the network structures. VE-cadherin also appears to be required for cell-cell association, but additionally appears to play some role in the process of vacuolization or vacuole fusion leading to intercellular lumen formation. | 10487846
 |
Cleavage of beta-catenin and plakoglobin and shedding of VE-cadherin during endothelial apoptosis: evidence for a role for caspases and metalloproteinases.
Herren, B; Levkau, B; Raines, EW; Ross, R
Molecular biology of the cell
9
1589-601
1998
Zobrazit abstrakt
Growth factor deprivation of endothelial cells induces apoptosis, which is characterized by membrane blebbing, cell rounding, and subsequent loss of cell-matrix and cell-cell contacts. In this study, we show that initiation of endothelial apoptosis correlates with cleavage and disassembly of intracellular and extracellular components of adherens junctions. beta-Catenin and plakoglobin, which form intracellular links between vascular endothelial cadherin (VE-cadherin) and actin-binding alpha-catenin in adherens junctions, are cleaved in apoptotic cells. In vitro incubations of cell lysates and immunoprecipitates with recombinant caspases indicate that CPP32 and Mch2 are involved, possibly by initiating proteolytic processing. Cleaved beta-catenin from lysates of apoptotic cells does not bind to endogenous alpha-catenin, whereas plakoglobin retains its binding capacity. The extracellular portion of the adherens junctions is also altered during apoptosis because VE-cadherin, which mediates endothelial cell-cell interactions, dramatically decreases on the surface of cells. An extracellular fragment of VE-cadherin can be detected in the conditioned medium, and this "shedding" of VE-cadherin can be blocked by an inhibitor of metalloproteinases. Thus, cleavage of beta-catenin and plakoglobin and shedding of VE-cadherin may act in concert to disrupt structural and signaling properties of adherens junctions and may actively interrupt extracellular signals required for endothelial cell survival. | 9614196
 |