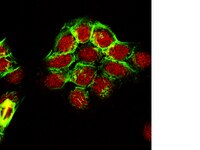
Perturbation of BRD4 protein function by BRD4-NUT protein abrogates cellular differentiation in NUT midline carcinoma.
Yan, J; Diaz, J; Jiao, J; Wang, R; You, J
The Journal of biological chemistry
286
27663-75
2010
Zobrazit abstrakt
NUT midline carcinoma (NMC) belongs to a class of highly lethal and poorly differentiated epithelial cancers arising mainly in human midline organs. NMC is caused by the chromosome translocation-mediated fusion of the NUT (nuclear protein in testis) gene on chromosome 15 to a few other genes, most frequently the BRD4 gene on chromosome 19. The mechanism by which the BRD4-NUT fusion product blocks NMC cellular differentiation and contributes to oncogenesis remains elusive. In this study, we show that BRD4-NUT and BRD4 colocalize in discrete nuclear foci that are hyperacetylated but transcriptionally inactive. BRD4-NUT recruits histone acetyltransferases to induce histone hyperacetylation in these chromatin foci, which provide docking sites for accumulation of additional BRD4 and associated P-TEFB (positive transcription elongation factor b) complexes in the transcriptionally inactive BRD4-NUT foci. These molecular events lead to repression of a BRD4·P-TEFB downstream target gene c-fos, a component of activator protein 1 (AP-1), that directly regulates epithelial differentiation. Knockdown of BRD4-NUT in NMC cells disperses the transcriptionally inactive chromatin foci and releases the transcriptional activators to stimulate c-fos expression, leading to restoration of cellular differentiation. Our study provides a novel mechanism by which the BRD4-NUT oncogene perturbs BRD4 functions to block cellular differentiation and to contribute to the oncogenic progression in the highly aggressive NMC. | 21652721
 |
GCN5: a supervisor in all-inclusive control of vertebrate cell cycle progression through transcription regulation of various cell cycle-related genes.
Kikuchi, Hidehiko, et al.
Gene, 347: 83-97 (2005)
2004
Zobrazit abstrakt
Histone acetyltransferases (HATs) are involved in the acetylation of core histones, which is an important event for transcription regulation through alterations in the chromatin structure in eukaryotes. To clarify participatory in vivo roles of two such enzymes known as GCN5 and PCAF, we generated homozygous DT40 mutants, DeltaGCN5 and DeltaPCAF, devoid of two alleles of each of the GCN5 and PCAF genes, respectively, with the help of gene targeting technique. While the PCAF-deficiency exhibited no effect on growth rate, the GCN5-deficiency caused delayed growth rate of DT40 cells. FACS analyses revealed not only that the number of cells in S phase decreased, but also that the cell cycle progression was suppressed at G1/S phase transition for DeltaGCN5. RT-PCR analyses revealed that the GCN5-deficiency exhibited opposite influences on transcriptions of G1/S phase transition-related genes, i.e. repressions for E2F-1, E2F-3, E2F-4, E2F-6, DP-2, cyclin A, cyclin D3, PCNA, cdc25B and p107; and activations for p27, c-myc, cyclin D2 and cyclin G1. Similarly, the deficiency influenced oppositely transcriptions of apoptosis-related genes, i.e. decreased expression of bcl-xL and increased expression of bcl-2. Immunoblotting analyses using a number of anti-acetylated histone antisera revealed that the GCN5-deficiency led to decreased acetylation levels of K16/H2B and K9/H3, and increased those of K7/H2A, K18/H3, K23/H3, K27/H3, K8/H4 and K12/H4. These results indicate that GCN5 preferentially acts as a supervisor in the normal cell cycle progression having comprehensive control over expressions of these cell cycle-related genes, as well as apoptosis-related genes, probably via alterations in the chromatin structure, mimicked by changing acetylation status of core histones, surrounding these widely distributed genes. | 15715965
 |
c-Myc transformation domain recruits the human STAGA complex and requires TRRAP and GCN5 acetylase activity for transcription activation.
Liu, Xiaohui, et al.
J. Biol. Chem., 278: 20405-12 (2003)
2003
Zobrazit abstrakt
Deregulation of the c-Myc oncoprotein (Myc) is implicated in many types of cancer. Myc is a sequence-specific transcription factor that regulates transcription of genes involved in the control of cell proliferation and apoptosis via mechanisms that are still poorly understood. Cell transformation by Myc involves its association with the transformation-transactivation domain-associated protein (TRRAP) and the human histone acetyltransferase (HAT) GCN5. TRRAP and GCN5 are components of a variety of shared and distinct multiprotein HAT complexes with diverse functions. Myc induces TRRAP recruitment and histone hyperacetylation at specific Myc-activated genes in vivo. However, the identity of the HAT complexes recruited by Myc and the roles of TRRAP and GCN5 in Myc function are still unclear. Here we show that Myc co-recruits TRRAP and GCN5 via direct physical interactions of its N-terminal activation/transformation domain with the human STAGA (SPT3-TAF-GCN5 acetylase) coactivator complex. We demonstrate that GCN5 and TRRAP cooperate to enhance transcription activation by the N-terminal activation domain of Myc in vivo and that this synergy requires both the SPT3/GCN5 interaction domain of TRRAP and the HAT activity of GCN5. Thus, TRRAP might function as an adaptor within the STAGA complex, which helps recruit GCN5 HAT activity to Myc during transcription activation. | 12660246
 |
Sir2 regulates skeletal muscle differentiation as a potential sensor of the redox state.
Fulco, Marcella, et al.
Mol. Cell, 12: 51-62 (2003)
2003
Zobrazit abstrakt
Sir2 is a NAD(+)-dependent histone deacetylase that controls gene silencing, cell cycle, DNA damage repair, and life span. Prompted by the observation that the [NAD(+)]/[NADH] ratio is subjected to dynamic fluctuations in skeletal muscle, we have tested whether Sir2 regulates muscle gene expression and differentiation. Sir2 forms a complex with the acetyltransferase PCAF and MyoD and, when overexpressed, retards muscle differentiation. Conversely, cells with decreased Sir2 differentiate prematurely. To inhibit myogenesis, Sir2 requires its NAD(+)-dependent deacetylase activity. The [NAD(+)]/[NADH] ratio decreases as muscle cells differentiate, while an increased [NAD(+)]/[NADH] ratio inhibits muscle gene expression. Cells with reduced Sir2 levels are less sensitive to the inhibition imposed by an elevated [NAD(+)]/[NADH] ratio. These results indicate that Sir2 regulates muscle gene expression and differentiation by possibly functioning as a redox sensor. In response to exercise, food intake, and starvation, Sir2 may sense modifications of the redox state and promptly modulate gene expression. | 12887892
 |
Regulation of HIV-1 gene expression by histone acetylation and factor recruitment at the LTR promoter.
Lusic, Marina, et al.
EMBO J., 22: 6550-61 (2003)
2003
Zobrazit abstrakt
In HIV-1 infected cells, the LTR promoter, once organized into chromatin, is transcriptionally inactive in the absence of stimulation. To examine the chromosomal events involved in transcriptional activation, we analyzed histone acetylation and factor recruitment at contiguous LTR regions by a quantitative chromatin immunoprecipitation assay. In chronically infected cells treated with a phorbol ester, we found that acetylation of both histones H3 and H4 occurs at discrete nucleosomal regions before the onset of viral mRNA transcription. Concomitantly, we observed the recruitment of known cellular acetyl-transferases to the promoter, including CBP, P/CAF and GCN5, as well as that of the p65 subunit of NF-kappa B. The specific contribution of the viral Tat transactivator was assayed in cells harboring the sole LTR. We again observed nucleosomal acetylation and the recruitment of specific co-factors to the viral LTR upon activation by either recombinant Tat or a phorbol ester. Strikingly, P/CAF was found associated with the promoter only in response to Tat. Taken together, these results contribute to the elucidation of the molecular events underlying HIV-1 transcriptional activation. | 14657027
 |