475838 Sigma-AldrichMifepristone - CAS 84371-65-3 - Calbiochem
A cell-permeable synthetic steroid that acts as a potent antagonist of progesterone and glucocorticoid receptors.
More>> A cell-permeable synthetic steroid that acts as a potent antagonist of progesterone and glucocorticoid receptors. Less<<Synonyms: (11β,17β)-11-(4-(Dimethylamino)phenyl)-17-hydroxy-17-(1-propynyl)estra-4,9-dien-3-one, RU-486
Recommended Products
Overview
| Replacement Information |
|---|
Key Spec Table
| CAS # | Empirical Formula |
|---|---|
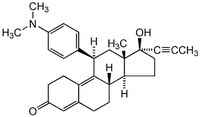
| 84371-65-3 | C₂₉H₃₅NO₂ |
Products
| Catalogue Number | Packaging | Qty/Pack | |
|---|---|---|---|
| 475838-50MG | Plastic ampoule | 50 mg |
| Product Information | |
|---|---|
| CAS number | 84371-65-3 |
| ATP Competitive | N |
| Form | Yellow solid |
| Hill Formula | C₂₉H₃₅NO₂ |
| Chemical formula | C₂₉H₃₅NO₂ |
| Reversible | N |
| Structure formula Image | |
| Quality Level | MQ100 |
| Applications |
|---|
| Biological Information | |
|---|---|
| Primary Target | Progesterone and glucocorticoid receptors |
| Purity | ≥99% by titration |
| Physicochemical Information | |
|---|---|
| Cell permeable | Y |
| Contaminants | Steroid substances: ≤0.1% |
| Dimensions |
|---|
| Materials Information |
|---|
| Toxicological Information |
|---|
| Safety Information according to GHS | |
|---|---|
| RTECS | KG2955000 |
| Product Usage Statements |
|---|
| Packaging Information | |
|---|---|
| Packaged under inert gas | Packaged under inert gas |
| Transport Information |
|---|
| Supplemental Information |
|---|
| Specifications |
|---|
| Global Trade Item Number | |
|---|---|
| Catalogue Number | GTIN |
| 475838-50MG | 04055977184976 |
Documentation
Mifepristone - CAS 84371-65-3 - Calbiochem SDS
| Title |
|---|
Mifepristone - CAS 84371-65-3 - Calbiochem Certificates of Analysis
| Title | Lot Number |
|---|---|
| 475838 |
References
| Reference overview |
|---|
| McCullers, D.L., et al. 2002. Neuroscience 109, 219. Sidell, N., et al. 2002. Ann. N. Y. Acad. Sci. 955, 159. Gao, F., et al. 2001. Acta Pharmacol. Sin. 22, 524. Hyder, S.M., et al. 2001. Int. J. Cancer 92, 469. Lam, F.C., et al. 2001. J. Neurochem. 76, 1121. Sridhar, S., et al. 2001. Cancer Res. 61, 7179. Behl, C., et al. 1997. Eur. J. Neurosci. 9, 912. Greb, R.R., et al. 1997. Hum. Reprod. 12, 1280. Gruol, D.J., et al. 1994. Cancer Res. 54, 3088. |
Brochure
| Title |
|---|
| Alzheimer's Disease Brochure & Technical Guide |