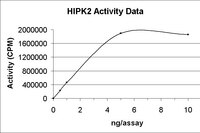
Homeodomain-interacting protein kinase 1 modulates Daxx localization, phosphorylation, and transcriptional activity.
Ecsedy, Jeffrey A, et al.
Mol. Cell. Biol., 23: 950-60 (2003)
2003
Pokaż streszczenie
We describe an interaction between homeodomain-interacting protein kinase 1 (HIPK1) and Daxx, two transcriptional regulators important in transducing growth-regulatory signals. We demonstrate that HIPK1 is ubiquitously expressed in mice and humans and localizes predominantly to the nucleus. Daxx normally resides within the nucleus in promyelocytic leukemia protein (PML) oncogenic domains (PODs), where it physically interacts with PML. Under certain circumstances, Daxx is relocalized from PODs to chromatin, where it then acts as a transcriptional repressor through an association with histone deacetylase (HDAC1). We propose two novel mechanisms for regulating the activity of Daxx, both mediated by HIPK1. First, HIPK1 physically interacts with Daxx in cells and consequently relocalizes Daxx from PODs. Daxx relocalization disrupts its interaction with PML and augments its interaction with HDAC1, likely influencing Daxx activity. Although the relocalization of Daxx from PODs is phosphorylation independent, an active HIPK1 kinase domain is required, suggesting that HIPK1 autophosphorylation is important in this interaction. Second, HIPK1 phosphorylates Daxx on Ser 669, and phosphorylation of this site is important in modulating the ability of Daxx to function as a transcriptional repressor. Mutation of Daxx Ser 669 to Ala results in increased repression in three of four transcriptional reporters, suggesting that phosphorylation by HIPK1 diminishes Daxx transcriptional repression of specific promoters. Taken together, our results indicate that HIPK1 and Daxx collaborate in regulating transcription. | 12529400
 |
Characterization of cells and gene-targeted mice deficient for the p53-binding kinase homeodomain-interacting protein kinase 1 (HIPK1).
Kondo, Seiji, et al.
Proc. Natl. Acad. Sci. U.S.A., 100: 5431-6 (2003)
2003
Pokaż streszczenie
The tumor suppressor p53 is regulated in part by binding to cellular proteins. We used p53 as bait in the yeast two-hybrid system and isolated homeodomain-interacting protein kinase 1 (HIPK1) as a p53-binding protein. Deletion analysis showed that amino acids 100-370 of p53 and amino acids 885-1093 of HIPK1 were sufficient for HIPK1-p53 interaction. HIPK1 was capable of autophosphorylation and specific serine phosphorylation of p53. The HIPK1 gene was highly expressed in human breast cancer cell lines and oncogenically transformed mouse embryonic fibroblasts. HIPK1 was localized to human chromosome band 1p13, a site frequently altered in cancers. Gene-targeted HIPK1-/- mice were grossly normal but oncogenically transformed HIPK1 -/- mouse embryonic fibroblasts exhibited reduced transcription of Mdm2 and were more susceptible than transformed HIPK1+/+ cells to apoptosis induced by DNA damage. Carcinogen-treated HIPK1 -/- mice developed fewer and smaller skin tumors than HIPK1+/+ mice. HIPK1 may thus play a role in tumorigenesis, perhaps by means of the regulation of p53 and/or Mdm2. | 12702766
 |
Role of the ASK1-SEK1-JNK1-HIPK1 signal in Daxx trafficking and ASK1 oligomerization.
Song, Jae J and Lee, Yong J
J. Biol. Chem., 278: 47245-52 (2003)
2003
Pokaż streszczenie
Overexpression of JNK binding domain inhibited glucose deprivation-induced JNK1 activation, relocalization of Daxx from the nucleus to the cytoplasm, and apoptosis signal-regulating kinase 1 (ASK1) oligomerization in human prostate adenocarcinoma DU-145 cells. However, SB203580, a p38 inhibitor, did not prevent relocalization of Daxx and oligomerization of ASK1 during glucose deprivation. Studies from in vivo labeling and immune complex kinase assay demonstrated that phosphorylation of Daxx occurred during glucose deprivation, and its phosphorylation was mediated through the ASK1-SEK1-JNK1-HIPK1 signal transduction pathway. Data from immunofluorescence staining and protein interaction assay suggest that phosphorylated Daxx may be translocated to the cytoplasm, bind to ASK1, and subsequently lead to ASK1 oligomerization. Mutation of Daxx Ser667 to Ala results in suppression of Daxx relocalization during glucose deprivation, suggesting that Ser667 residue plays an important role in the relocalization of Daxx. Unlike wild-type Daxx, a Daxx deletion mutant (amino acids 501-625) mainly localized to the cytoplasm, where it associated with ASK1, activated JNK1, and induced ASK1 oligomerization without glucose deprivation. Taken together, these results show that glucose deprivation activates the ASK1-SEK1-JNK1-HIPK1 pathway, and the activated HIPK1 is probably involved in the relocalization of Daxx from the nucleus to the cytoplasm. The relocalized Daxx may play an important role in glucose deprivation-induced ASK1 oligomerization. | 12968034
 |
Homeodomain-interacting protein kinases, a novel family of co-repressors for homeodomain transcription factors.
Kim, Y H, et al.
J. Biol. Chem., 273: 25875-9 (1998)
1998
Pokaż streszczenie
A novel family of cofactors that differentially interact with homeoproteins have been identified via a yeast two-hybrid screen. The proteins contain a conserved protein kinase domain that is separated from a domain that interacts with homeoproteins and hence are termed homeodomain-interacting protein kinases (HIPKs): HIPK1, HIPK2, and HIPK3. We show that HIPKs are nuclear kinases using GFP-HIPK fusion constructs. The DNA binding activity of the NK-3 homeoprotein is greatly enhanced by HIPK2, but this effect is independent of its phosphorylation by HIPK2. In cultured cells, HIPKs localize to nuclear speckles and potentiate the repressor activities of NK homeoproteins. The co-repressor activity of HIPKs depends on both its homeodomain interaction domain and a co-repressor domain that maps to the N terminus. Thus, HIPKs represent a heretofore undescribed family of co-repressors for homeodomain transcription factors. | 9748262
 |