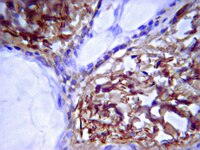
Development of the follicular basement membrane during human gametogenesis and early folliculogenesis.
Heeren, AM; van Iperen, L; Klootwijk, DB; de Melo Bernardo, A; Roost, MS; Gomes Fernandes, MM; Louwe, LA; Hilders, CG; Helmerhorst, FM; van der Westerlaken, LA; Chuva de Sousa Lopes, SM
BMC developmental biology
15
4
2015
Pokaż streszczenie
In society, there is a clear need to improve the success rate of techniques to restore fertility. Therefore a deeper knowledge of the dynamics of the complex molecular environment that regulates human gametogenesis and (early) folliculogenesis in vivo is necessary. Here, we have studied these processes focusing on the formation of the follicular basement membrane (BM) in vivo.The distribution of the main components of the extracellular matrix (ECM) collagen IV, laminin and fibronectin by week 10 of gestation (W10) in the ovarian cortex revealed the existence of ovarian cords and of a distinct mesenchymal compartment, resembling the organization in the male gonads. By W17, the first primordial follicles were assembled individually in that (cortical) mesenchymal compartment and were already encapsulated by a BM of collagen IV and laminin, but not fibronectin. In adults, in the primary and secondary follicles, collagen IV, laminin and to a lesser extent fibronectin were prominent in the follicular BM.The ECM-molecular niche compartimentalizes the female gonads from the time of germ cell colonization until adulthood. This knowledge may contribute to improve methods to recreate the environment needed for successful folliculogenesis in vitro and that would benefit a large number of infertility patients. | | 25605128
 |
Glycosaminoglycan-based hydrogels to modulate heterocellular communication in in vitro angiogenesis models.
Chwalek, K; Tsurkan, MV; Freudenberg, U; Werner, C
Scientific reports
4
4414
2014
Pokaż streszczenie
Angiogenesis, the outgrowth of blood vessels, is crucial in development, disease and regeneration. Studying angiogenesis in vitro remains challenging because the capillary morphogenesis of endothelial cells (ECs) is controlled by multiple exogenous signals. Therefore, a set of in situ-forming starPEG-heparin hydrogels was used to identify matrix parameters and cellular interactions that best support EC morphogenesis. We showed that a particular type of soft, matrix metalloproteinase-degradable hydrogel containing covalently bound integrin ligands and reversibly conjugated pro-angiogenic growth factors could boost the development of highly branched, interconnected, and lumenized endothelial capillary networks. Using these effective matrix conditions, 3D heterocellular interactions of ECs with different mural cells were demonstrated that enabled EC network modulation and maintenance of stable vascular capillaries over periods of about one month in vitro. The approach was also shown to permit in vitro tumor vascularization experiments with unprecedented levels of control over both ECs and tumor cells. In total, the introduced 3D hydrogel co-culture system could offer unique options for dissecting and adjusting biochemical, biophysical, and cell-cell triggers in tissue-related vascularization models. | | 24643064
 |
Lymphangiogenesis and angiogenesis during human fetal pancreas development.
Roost, MS; van Iperen, L; de Melo Bernardo, A; Mummery, CL; Carlotti, F; de Koning, EJ; Chuva de Sousa Lopes, SM
Vascular cell
6
22
2014
Pokaż streszczenie
The complex endocrine and exocrine functionality of the human pancreas depends on an efficient fluid transport through the blood and the lymphatic vascular systems. The lymphatic vasculature has key roles in the physiology of the pancreas and in regulating the immune response, both important for developing successful transplantation and cell-replacement therapies to treat diabetes. However, little is known about how the lymphatic and blood systems develop in humans. Here, we investigated the establishment of these two vascular systems in human pancreas organogenesis in order to understand neovascularization in the context of emerging regenerative therapies.We examined angiogenesis and lymphangiogenesis during human pancreas development between 9 and 22 weeks of gestation (W9-W22) by immunohistochemistry.As early as W9, the peri-pancreatic mesenchyme was populated by CD31-expressing blood vessels as well as LYVE1- and PDPN-expressing lymphatic vessels. The appearance of smooth muscle cell-coated blood vessels in the intra-pancreatic mesenchyme occurred only several weeks later and from W14.5 onwards the islets of Langerhans also became heavily irrigated by blood vessels. In contrast to blood vessels, LYVE1- and PDPN-expressing lymphatic vessels were restricted to the peri-pancreatic mesenchyme until later in development (W14.5-W17), and some of these invading lymphatic vessels contained smooth muscle cells at W17. Interestingly, between W11-W22, most large caliber lymphatic vessels were lined with a characteristic, discontinuous, collagen type IV-rich basement membrane. Whilst lymphatic vessels did not directly intrude the islets of Langerhans, three-dimensional reconstruction revealed that they were present in the vicinity of islets of Langerhans between W17-W22.Our data suggest that the blood and lymphatic machinery in the human pancreas is in place to support endocrine function from W17-W22 onwards. Our study provides the first systematic assessment of the progression of lymphangiogenesis during human pancreatic development. | | 25785186
 |
Spatial and temporal analysis of extracellular matrix proteins in the developing murine heart: a blueprint for regeneration.
Hanson, KP; Jung, JP; Tran, QA; Hsu, SP; Iida, R; Ajeti, V; Campagnola, PJ; Eliceiri, KW; Squirrell, JM; Lyons, GE; Ogle, BM
Tissue engineering. Part A
19
1132-43
2013
Pokaż streszczenie
The extracellular matrix (ECM) of the embryonic heart guides assembly and maturation of cardiac cell types and, thus, may serve as a useful template, or blueprint, for fabrication of scaffolds for cardiac tissue engineering. Surprisingly, characterization of the ECM with cardiac development is scattered and fails to comprehensively reflect the spatiotemporal dynamics making it difficult to apply to tissue engineering efforts. The objective of this work was to define a blueprint of the spatiotemporal organization, localization, and relative amount of the four essential ECM proteins, collagen types I and IV (COLI, COLIV), elastin (ELN), and fibronectin (FN) in the left ventricle of the murine heart at embryonic stages E12.5, E14.5, and E16.5 and 2 days postnatal (P2). Second harmonic generation (SHG) imaging identified fibrillar collagens at E14.5, with an increasing density over time. Subsequently, immunohistochemistry (IHC) was used to compare the spatial distribution, organization, and relative amounts of each ECM protein. COLIV was found throughout the developing heart, progressing in amount and organization from E12.5 to P2. The amount of COLI was greatest at E12.5 particularly within the epicardium. For all stages, FN was present in the epicardium, with highest levels at E12.5 and present in the myocardium and the endocardium at relatively constant levels at all time points. ELN remained relatively constant in appearance and amount throughout the developmental stages except for a transient increase at E16.5. Expression of ECM mRNA was determined using quantitative polymerase chain reaction and allowed for comparison of amounts of ECM molecules at each time point. Generally, COLI and COLIII mRNA expression levels were comparatively high, while COLIV, laminin, and FN were expressed at intermediate levels throughout the time period studied. Interestingly, levels of ELN mRNA were relatively low at early time points (E12.5), but increased significantly by P2. Thus, we identified changes in the spatial and temporal localization of the primary ECM of the developing ventricle. This characterization can serve as a blueprint for fabrication techniques, which we illustrate by using multiphoton excitation photochemistry to create a synthetic scaffold based on COLIV organization at P2. Similarly, fabricated scaffolds generated using ECM components, could be utilized for ventricular repair. | Immunohistochemistry | 23273220
 |
Nf1 limits epicardial derivative expansion by regulating epithelial to mesenchymal transition and proliferation.
Baek, ST; Tallquist, MD
Development (Cambridge, England)
139
2040-9
2011
Pokaż streszczenie
The epicardium is the primary source of coronary vascular smooth muscle cells (cVSMCs) and fibroblasts that reside in the compact myocardium. To form these epicardial-derived cells (EPDCs), the epicardium undergoes the process of epithelial to mesenchymal transition (EMT). Although several signaling pathways have been identified that disrupt EMT, no pathway has been reported that restricts this developmental process. Here, we identify neurofibromin 1 (Nf1) as a key mediator of epicardial EMT. To determine the function of Nf1 during epicardial EMT and the formation of epicardial derivatives, cardiac fibroblasts and cVSMCs, we generated mice with a tissue-specific deletion of Nf1 in the epicardium. We found that mutant epicardial cells transitioned more readily to mesenchymal cells in vitro and in vivo. The mesothelial epicardium lost epithelial gene expression and became more invasive. Using lineage tracing of EPDCs, we found that the process of EMT occurred earlier in Nf1 mutant hearts, with an increase in epicardial cells entering the compact myocardium. Moreover, loss of Nf1 caused increased EPDC proliferation and resulted in more cardiac fibroblasts and cVSMCs. Finally, we were able to partially reverse the excessive EMT caused by loss of Nf1 by disrupting Pdgfrα expression in the epicardium. Conversely, Nf1 activation was able to inhibit PDGF-induced epicardial EMT. Our results demonstrate a regulatory role for Nf1 during epicardial EMT and provide insights into the susceptibility of patients with disrupted NF1 signaling to cardiovascular disease. | Immunohistochemistry | 22535408
 |
Clinical and laboratory features of neuropathies with serum IgM binding to TS-HDS.
Alan Pestronk,Robert E Schmidt,Rati M Choksi,R Brian Sommerville,Muhammad T Al-Lozi
Muscle & nerve
45
2011
Pokaż streszczenie
In this investigation we studied clinical and laboratory features of polyneuropathies in patients with serum IgM binding to the trisulfated disaccharide IdoA2S-GlcNS-6S (TS-HDS). | | 22581541
 |
Leptospira interrogans binds to human cell surface receptors including proteoglycans.
Breiner, DD; Fahey, M; Salvador, R; Novakova, J; Coburn, J
Infection and immunity
77
5528-36
2009
Pokaż streszczenie
Leptospirosis is a global public health problem, primarily in the tropical developing world. The pathogenic mechanisms of the causative agents, several members of the genus Leptospira, have been underinvestigated. The exception to this trend has been the demonstration of the binding of pathogenic leptospires to the extracellular matrix (ECM) and its components. In this work, interactions of Leptospira interrogans bacteria with mammalian cells, rather than the ECM, were examined. The bacteria bound more efficiently to the cells than to the ECM, and a portion of this cell-binding activity was attributable to attachment to glycosaminoglycan (GAG) chains of proteoglycans (PGs). Chondroitin sulfate B PGs appeared to be the primary targets of L. interrogans attachment, while heparan sulfate PGs were much less important. Inhibition of GAG/PG-mediated attachment resulted in partial inhibition of bacterial attachment, suggesting that additional receptors for L. interrogans await identification. GAG binding may participate in the pathogenesis of leptospirosis within the host animal. In addition, because GAGs are expressed on the luminal aspects of epithelial cells in the proximal tubules of the kidneys, this activity may play a role in targeting the bacteria to this critical site. Because GAGs are shed in the urine, GAG binding may also be important for transmission to new hosts through the environment. Pełny tekst artykułu | Western Blotting | 19805539
 |
Formation of smooth muscle alpha actin filaments in CD34+ bone marrow cells on arterial elastic laminae: potential role of SH2 domain-containing protein tyrosine phosphatase-1.
Shu Q Liu, Brandon J Tefft, Andy Zhang, Li-Qun Zhang, Yu H Wu
Matrix biology : journal of the International Society for Matrix Biology
27
282-94
2008
Pokaż streszczenie
Arterial smooth muscle cells (SMCs) are present in the elastic lamina-containing media, suggesting that the elastic laminae may regulate the development of SMCs. Here, we investigated the role of elastic laminae in regulating the formation of SM alpha actin filaments in mouse CD34+ bone marrow cells and the role of a protein tyrosine phosphatase, SH2 domain-containing protein tyrosine phosphatase (SHP)-1, in the mediation of this process. Mouse CD34+ bone marrow cells were isolated by magnetic separation and used for assessing the influence of elastic laminae and collagen matrix on the formation of SM alpha actin filaments. CD34+ cells with transgenic SHP-1 knockout or siRNA-mediated SHP-1 knockdown were used to assess the role of SHP-1 in mediating the formation of SM alpha actin filaments. In cell culture tests, elastic laminae, but not collagen matrix, stimulated the formation of SM alpha actin filaments in CD34+ cells. The phosphatase SHP-1 mediated the stimulatory effect of elastic laminae. The interaction of CD34+ cells with elastic laminae, but not with collagen matrix, induced activation of SHP-1. The suppression of SHP-1 by transgenic SHP-1 knockout or siRNA-mediated SHP-1 knockdown significantly reduced the formation of SM alpha actin filaments in CD34+ cells cultured on elastic laminae. The in vitro observations were confirmed by using an in vivo model of implantation of elastic lamina and collagen matrix scaffolds into the aorta. These observations suggest that elastic laminae stimulate the formation of SM alpha actin filaments in CD34+ bone marrow cells and SHP-1 mediates the stimulatory effect of elastic laminae. | | 18258420
 |
Ullrich myopathy phenotype with secondary ColVI defect identified by confocal imaging and electron microscopy analysis.
Stefania Petrini,Adele D'Amico,Patrizio Sale,Laura Lucarini,Patrizia Sabatelli,Alessandra Tessa,Betti Giusti,Margherita Verardo,Rosalba Carrozzo,Elisabetta Mattioli,Marina Scarpelli,Mon-Li Chu,Guglielmina Pepe,Matteo Antonio Russo,Enrico Bertini
Neuromuscular disorders : NMD
17
2007
Pokaż streszczenie
Ullrich congenital muscular dystrophy (UCMD) is clinically characterized by muscle weakness, proximal contractures and distal hyperlaxity and morphologically branded by absence or reduction of collagen VI (ColVI), in muscle and in cultured fibroblasts. The ColVI defect is generally related to COL6 genes mutations, however UCDM patients without COL6 mutations have been recently reported, suggesting genetic heterogeneity. We report comparative morphological findings between a UCMD patient harboring a homozygous COL6A2 mutation and a patient with a typical UCMD phenotype in which mutations in COL6 genes were excluded. The patient with no mutations in COL6 genes exhibited a partial ColVI defect, which was only detected close to the basal membrane of myofibers. We describe how confocal microscopy and rotary-shadowing electron microscopy may be useful to identify a secondary ColVI defect. | | 17588753
 |
Transgenic engineering of male-specific muscular hypertrophy.
Pirottin, D; Grobet, L; Adamantidis, A; Farnir, F; Herens, C; Schrøder, HD; Georges, M
Proceedings of the National Academy of Sciences of the United States of America
102
6413-8
2004
Pokaż streszczenie
Using a two-step procedure involving insertional gene targeting and recombinase-mediated cassette exchange in ES cells, we have produced two lines of transgenic mice expressing a dominant-negative latency-associated myostatin propeptide under control of the myosin light chain 1F promoter and 1/3 enhancer from the TSPY locus on the Y chromosome. Males of the corresponding lines are characterized by a 5-20% increase in skeletal muscle mass. This experiment demonstrates the feasibility of a more efficient cattle production system combining superior beef production abilities for bulls and dairy abilities for cows. Pełny tekst artykułu | | 15851664
 |


















-ethanol[840111_(Ethylmercapto)-ethanol-ALL].jpg)