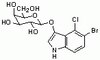
MicroRNAs are essential for stretch-induced vascular smooth muscle contractile differentiation via microRNA (miR)-145-dependent expression of L-type calcium channels.
Turczynska, KM; Sadegh, MK; Hellstrand, P; Swärd, K; Albinsson, S
The Journal of biological chemistry
287
19199-206
2011
Pokaż streszczenie
Stretch of the vascular wall is an important stimulus to maintain smooth muscle contractile differentiation that is known to depend on L-type calcium influx, Rho-activation, and actin polymerization. The role of microRNAs in this response was investigated using tamoxifen-inducible and smooth muscle-specific Dicer KO mice. In the absence of Dicer, which is required for microRNA maturation, smooth muscle microRNAs were completely ablated. Stretch-induced contractile differentiation and Rho-dependent cofilin-2 phosphorylation were dramatically reduced in Dicer KO vessels. On the other hand, acute stretch-sensitive growth signaling, which is independent of influx through L-type calcium channels, was not affected by Dicer KO. Contractile differentiation induced by the actin polymerizing agent jasplakinolide was not altered by deletion of Dicer, suggesting an effect upstream of actin polymerization. Basal and stretch-induced L-type calcium channel expressions were both decreased in Dicer KO portal veins, and inhibition of L-type channels in control vessels mimicked the effects of Dicer deletion. Furthermore, inhibition of miR-145, a highly expressed microRNA in smooth muscle, resulted in a similar reduction of L-type calcium channel expression. This was abolished by the Ca(2+)/calmodulin-dependent protein kinase II inhibitor KN93, suggesting that Ca(2+)/calmodulin-dependent protein kinase IIδ, a target of miR-145 and up-regulated in Dicer KO, plays a role in the regulation of L-type channel expression. These results show that microRNAs play a crucial role in stretch-induced contractile differentiation in the vascular wall in part via miR-145-dependent regulation of L-type calcium channels. | 22474293
 |
Nitric oxide inhibits endothelin-1-induced neonatal cardiomyocyte hypertrophy via a RhoA-ROCK-dependent pathway.
Hunter, J Craig, et al.
J. Mol. Cell. Cardiol., 47: 810-8 (2009)
2009
Pokaż streszczenie
Although nitric oxide (NO) has received extensive attention as an anti-hypertrophic agent the mechanisms underlying its regulation of endothelin-1 (ET-1) have not been fully elucidated. Since RhoA has been identified as an important mediator of cardiac hypertrophy and is inhibited by NO in vascular tissue, we sought to determine whether the anti-ET-1 effects of NO in cardiomyocytes were mediated via inhibition of the RhoA-ROCK cascade in the context of cardiac hypertrophy. Neonatal rat ventricular myocytes were cultured in the presence of ET-1 (10 nM) with or without pre-treatment with the NO donor S-nitroso-n-acetylpenicillamine (SNAP; 100 microM), 8-Br-cGMP (cGMP; 100 microM), the RhoA inhibitor C3 exoenzyme (C3; 30 ng/ml), or the ROCK inhibitor Y-27632 (10 microM). ET-1-induced cardiomyocyte hypertrophy was prevented by pre-treatment with SNAP, cGMP, C3, or Y-27632. The hypertrophic response to ET-1 was associated with significantly increased gene and protein expression of both NOS2 and NOS1 although NOS3 was unaffected. ET-1 treatment for 15 min increased membrane-bound RhoA 2.6-fold (p<0.05), which was prevented by both SNAP and cGMP (p<0.05). These effects were associated with a complete abrogation of ET-1-induced phosphorylation of the downstream target of RhoA, cofilin-2, that was mimicked by direct inhibition of RhoA and ROCK. In addition, confocal microscopy and Western blotting revealed that 24 h ET-1 treatment reduced the G- to F-actin ratio 67% (p<0.05) which was prevented by SNAP, cGMP, C3 and Y (p<0.05). Taken together, these results suggest that the anti-hypertrophic effects of NO are due, in part, to cGMP-dependent inhibition of the RhoA-ROCK-cofilin signalling pathway. These findings may be important in understanding the mechanisms of anti-ET-1 and anti-hypertrophic effects of NO as well as in the development of novel RhoA-targeted therapeutic interventions for treating cardiac hypertrophy. | 19799911
 |
Differential dependence of stretch and shear stress signaling on caveolin-1 in the vascular wall.
Albinsson, S; Nordström, I; Swärd, K; Hellstrand, P
American journal of physiology. Cell physiology
294
C271-9
2008
Pokaż streszczenie
The role of caveolae in stretch- versus flow-induced vascular responses was investigated using caveolin 1-deficient [knockout (KO)] mice. Portal veins were stretched longitudinally for 5 min (acute) or 72 h (organ culture). Basal ERK1/2 and Akt phosphorylation were increased in organ-cultured KO veins, as were protein synthesis and vessel wall cross sections. Stretch stimulated acute phosphorylation of ERK1/2 and long-term phosphorylation of focal adhesion kinase (FAK) and cofilin but did not affect Akt phosphorylation. Protein synthesis, and particularly synthesis of smooth muscle differentiation markers, was increased by stretch. These effects did not differ in portal veins from KO and control mice, which also showed the same contractile response to membrane depolarization and inhibition by the Rho kinase inhibitor Y-27632. KO carotid arteries had increased wall cross sections and responded to pressurization (120 mmHg) for 1 h with increased ERK1/2 but not Akt phosphorylation, similar to control arteries. Shear stress by flow for 15 min, on the other hand, increased phosphorylation of Akt in carotids from control but not KO mice. In conclusion, caveolin 1 contributes to low basal ERK1/2 and Akt activity and is required for Akt-dependent signals in response to shear stress (flow) but is not essential for trophic effects of stretch (pressure) in the vascular wall. | 17989209
 |
Stromal cell-derived factor 1alpha activates LIM kinase 1 and induces cofilin phosphorylation for T-cell chemotaxis.
Nishita, Michiru, et al.
Mol. Cell. Biol., 22: 774-83 (2002)
2002
Pokaż streszczenie
Stromal cell-derived factor 1 alpha (SDF-1alpha), the ligand for G-protein-coupled receptor CXCR4, is a chemotactic factor for T lymphocytes. LIM kinase 1 (LIMK1) phosphorylates cofilin, an actin-depolymerizing and -severing protein, at Ser-3 and regulates actin reorganization. We investigated the role of cofilin phosphorylation by LIMK1 in SDF-1alpha-induced chemotaxis of T lymphocytes. SDF-1alpha significantly induced the activation of LIMK1 in Jurkat human leukemic T cells and peripheral blood lymphocytes. SDF-1alpha also induced cofilin phosphorylation, actin reorganization, and activation of small GTPases, Rho, Rac, and Cdc42, in Jurkat cells. Pretreatment with pertussis toxin inhibited SDF-1alpha-induced LIMK1 activation, thus indicating that Gi protein is involved in LIMK1 activation. Expression of dominant negative Rac (DN-Rac), but not DN-Rho or DN-Cdc42, blocked SDF-1alpha-induced activation of LIMK1, which means that SDF-1alpha-induced LIMK1 activation is mediated by Rac but not by Rho or Cdc42. We used a cell-permeable peptide (S3 peptide) that contains the phosphorylation site (Ser-3) of cofilin to inhibit the cellular function of LIMK1. S3 peptide inhibited the kinase activity of LIMK1 in vitro. Treatment of Jurkat cells with S3 peptide inhibited the SDF-1alpha-induced cofilin phosphorylation, actin reorganization, and chemotactic response of Jurkat cells. These results suggest that the phosphorylation of cofilin by LIMK1 plays a critical role in the SDF-1alpha-induced chemotactic response of T lymphocytes. | 11784854
 |
Mitosis-dependent phosphorylation and activation of LIM-kinase 1.
Sumi, Tomoyuki, et al.
Biochem. Biophys. Res. Commun., 290: 1315-20 (2002)
2002
Pokaż streszczenie
LIM-kinases (LIMK1 and LIMK2) regulate actin cytoskeletal reorganization through phosphorylation of cofilin, an actin-depolymerizing factor of actin filaments. Here, we describe a detailed analysis of the cell-cycle-dependent activity of endogenous LIMK1. When HeLa cells were synchronized at prometaphase by nocodazole-treatment, LIMK1 was hyperphosphorylated, and its activity toward cofilin phosphorylation was markedly increased. During cell cycle progression, LIMK1 activity was low in interphase but reached a maximal level during mitosis. Activation of LIMK1 during mitosis was abrogated by roscovitine, a specific inhibitor of cyclin-dependent kinases (CDKs), suggesting that activation of CDKs directly or indirectly participates in LIMK1 activation. These results strongly suggest that LIMK1 may play an important role in the cell cycle progression through regulation of actin cytoskeletal rearrangements. | 11812007
 |