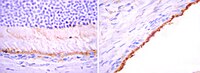
Attenuation of choroidal neovascularization by β(2)-adrenoreceptor antagonism.
Lavine, JA; Sang, Y; Wang, S; Ip, MS; Sheibani, N
JAMA ophthalmology
131
376-82
2013
Pokaż streszczenie
To determine whether β-adrenergic blockade inhibits choroidal neovascularization (CNV) in a mouse model of laser-induced CNV and to investigate the mechanism by which β-adrenoreceptor antagonism blunts CNV.Mice were subjected to laser burns, inducing CNV, and were treated with daily intraperitoneal injections of propranolol hydrochloride. Neovascularization was measured on choroidal-scleral flat mounts using intercellular adhesion molecule 2 immunofluorescence staining. The effect of β-adrenoreceptor signaling on expression of vascular endothelial growth factor (VEGF) was investigated using primary mouse choroidal endothelial cells (ChECs) and retinal pigment epithelial (RPE) cells. These cells were incubated with β-adrenoreceptor agonists and/or antagonists and assayed for Vegf messenger RNA and protein levels.University of Wisconsin School of Medicine and Public Health.Wild-type 6-week-old female C57BL/6j mice.Inhibition of CNV after propranolol treatment and Vegf messenger RNA and protein expression after treatment with β-adrenoreceptor agonists and antagonists.Propranolol-treated mice demonstrated a 50% reduction in laser-induced CNV. Treatment with norepinephrine bitartrate stimulated Vegf messenger RNA expression and protein secretion in ChECs and RPE cells. This effect was blocked by β2-adrenoreceptor antagonism and mimicked by β2-adrenoreceptor agonists.Attenuation of CNV is achieved by β-adrenergic blockade. The β2-adrenoreceptors regulate VEGF expression in ChECs and RPE cells.Antagonists of β-adrenoreceptors are safe and well tolerated in patients with glaucoma and cardiovascular disease. Thus, blockade of β-adrenoreceptors may provide a new avenue to inhibit VEGF expression in CNV. | 23303344
 |
Transplantation of human central nervous system stem cells - neuroprotection in retinal degeneration.
Trevor J McGill,Benjamin Cottam,Bin Lu,Shaomei Wang,Sergej Girman,Chunyu Tian,Stephen L Huhn,Ray D Lund,Alexandra Capela
The European journal of neuroscience
35
2011
Pokaż streszczenie
Stem cells derived from the human brain and grown as neurospheres (HuCNS-SC) have been shown to be effective in treating central neurodegenerative conditions in a variety of animal models. Human safety data in neurodegenerative disorders are currently being accrued. In the present study, we explored the efficacy of HuCNS-SC in a rodent model of retinal degeneration, the Royal College of Surgeons (RCS) rat, and extended our previous cell transplantation studies to include an in-depth examination of donor cell behavior and phenotype post-transplantation. As a first step, we have shown that HuCNS-SC protect host photoreceptors and preserve visual function after transplantation into the subretinal space of postnatal day 21 RCS rats. Moreover, cone photoreceptor density remained relatively constant over several months, consistent with the sustained visual acuity and luminance sensitivity functional outcomes. The novel findings of this study include the characterization and quantification of donor cell radial migration from the injection site and within the subretinal space as well as the demonstration that donor cells maintain an immature phenotype throughout the 7 months of the experiment and undergo very limited proliferation with no evidence of uncontrolled growth or tumor-like formation. Given the efficacy findings and lack of adverse events in the RCS rat in combination with the results from ongoing clinical investigations, HuCNS-SC appear to be a well-suited candidate for cell therapy in retinal degenerative conditions. | 22277045
 |