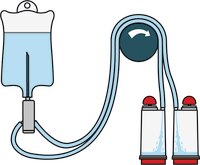
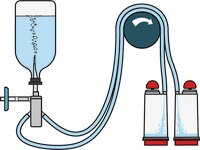
Steritest™ NEO Devices for Antibiotics and Products with Antimicrobial Activity
Red canister base with specific drain design. Low binding PVDF Durapore® membrane. This optimizes the rinsing of products that inhibit microbial growth. Canisters material: SAN
More
Red canister base with specific drain design. Low binding PVDF Durapore® membrane. This optimizes the rinsing of products that inhibit microbial growth. Canisters material: SAN Less
More>>
Less<<

Recommended Products
-

17-10241 Sigma-Aldrich ChIPAb+™ Acetyl-Histone H3 (Lys9/18) - ChIP Validated Antibody and Primer Set -

OP60 Sigma-Aldrich Anti-Bcl-2 (Ab-1) Mouse mAb (100) -

1005990001 Supelco Chlorine Test -

543900 Supelco Dimethyl sulfoxide -

104507 Millipore myo-Inositol -
820473 Sigma-Aldrich Probumin® Bovine Serum Albumin Biotech Grade, Powder -

3710-50GM Millipore OmniPur® Dextran Sulfate Sodium - CAS 9011-18-1 - Calbiochem -

MABN2678-25UG Sigma-Aldrich Anti-phospho-α-synuclein (Ser129) Antibody, clone PS129 -

500649 Sigma-Aldrich HMTase Inhibitor IX, MM-102 - Calbiochem -

06-734-25UG Sigma-Aldrich Anti-b-Catenin (rabbit polyclonal IgG)
Overview
Specifications
Ordering Information
Documentation
References
| Reference overview | Pub Med ID |
|---|---|
| The Effect of Vaporous Phase Hydrogen Peroxide on Sterility Test Devices Roche Lentine, Kerry and Robert J. Keller; Pharmaceutical Technology Europe, February 2003: 31- 42. Pharmaceutical Technology Europe, February 2003: 31- 42. 2003 | The Effect of Vaporous Phase Hydrogen Peroxide on Sterility Test Devices
 |
Related Products & Applications
Product Families
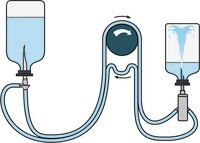
Steritest™ NEO AccessoriesFor faster, easier and safer sterility testing.Learn More >> |

Steritest™ NEO Device for products requiring increased Chemical CompatibilityGreen canister base with specific drain design. Low binding PVDF Durapore® membrane optimize the rinsing of products that inhibit microbial growth. Canisters material: polyamide polymer.Learn More >> |

Steritest™ NEO Devices for Products Without Antimicrobial AgentsBlue canister base. Mixed esters of cellulose membrane. This membrane provides an optimal filtration flow rate for standard products. Canisters material: SAN (Styrene-Acrylo Nitrile)Learn More >> |

Sterility TestingCountless configurationsLearn More >> |

Steritest™ EZ Devices Double-PackedIncrease your test method reliability with our gamma sterilized sterility testing deviceLearn More >> |
Related Products By: Application Facete
| Sterility Testing |
Related Products By: Brand Facete
| Steridilutor® |
| Steritest™ |
Categories
| Industrial Microbiology > Sterility Testing > Membrane Filtration Sterility Test > Steritest™ NEO Devices for Antibiotics & Products with Antimicrobial Activity |
Liquids in Ampoules
TZHVAB210 & TZHVAB205 (double packed)
- Single needle for easy access to ampoules
- Separate vent needle
Liquids in Collapsible Bags
TZHVAB210 & TZHVAB205 (double packed)
- Single needle for easy access to collapsible bags
- Separate vent needle
Liquids in Large Vials
TZHVLV210 & TZHVLV205 (double packed)
- Vented double needle for large glass containers with septa
Liquids in Small Vials
TZHVSV210 & TZHVSV205 (double packed)
- Vented double needle for small vials with septa
Soluble Powders in Vials
TZHVDV210 & TZHVDV205 (double packed)
- Double needles for small vials with septa
- Vented double needle
- Simultaneously dissolves/dilutes the sample in sterile diluent and transfers the resulting solution to canisters
Soluble Powders in Ampoules
TZHVDA210
- Double needles for small vials with septa
- Vented double needle
- Simultaneously dissolves/dilutes the sample in sterile diluent and transfers the resulting solution to canisters
Medical Devices and Collapsible Bags
TZHVMD210
- Three adapters provided; male Luer, female Luer or single needle allow connection to a variety of test devices
- Separate vent needle
Powders and Superpotent Antibiotics
TZVC00010 + TZHVAB210 or TZHVAB205 (double packed)
- Tubing and needle assembly for antibiotics and products containing antimicrobial activity that require dilution or dissolution
- Aseptically connects the diluent or dissolution fluid to the product container for dilution
- Used for pooling superpotent antibiotics to reduce product membrane contact time when product is then filtered
- Contains vent with expansion chamber for optimized venting
- Diluted product subsequently filtered with Steritest™ NEO device (TZHVAB210)