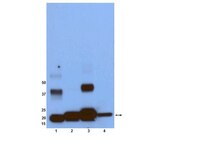
Site-specific processing of Ras and Rap1 Switch I by a MARTX toxin effector domain.
Antic, I; Biancucci, M; Zhu, Y; Gius, DR; Satchell, KJ
Nature communications
6
7396
2015
Abstract anzeigen
Ras (Rat sarcoma) protein is a central regulator of cell growth and proliferation. Mutations in the RAS gene are known to occur in human cancers and have been shown to contribute to carcinogenesis. In this study, we show that the multifunctional-autoprocessing repeats-in-toxin (MARTX) toxin-effector domain DUF5(Vv) from Vibrio vulnificus to be a site-specific endopeptidase that cleaves within the Switch 1 region of Ras and Rap1. DUF5(Vv) processing of Ras, which occurs both biochemically and in mammalian cell culture, inactivates ERK1/2, thereby inhibiting cell proliferation. The ability to cleave Ras and Rap1 is shared by DUF5(Vv) homologues found in other bacteria. In addition, DUF5(Vv )can cleave all Ras isoforms and KRas with mutations commonly implicated in malignancies. Therefore, we speculate that this new family of Ras/Rap1-specific endopeptidases (RRSPs) has potential to inactivate both wild-type and mutant Ras proteins expressed in malignancies. | | 26051945
 |
EGF-Induced Acetylation of Heterogeneous Nuclear Ribonucleoproteins Is Dependent on KRAS Mutational Status in Colorectal Cancer Cells.
Roda, D; Castillo, J; Telechea-Fernández, M; Gil, A; López-Rodas, G; Franco, L; González-Rodríguez, P; Roselló, S; Pérez-Fidalgo, JA; García-Trevijano, ER; Cervantes, A; Zaragozá, R
PloS one
10
e0130543
2015
Abstract anzeigen
KRAS mutational status is considered a negative predictive marker of the response to anti-EGFR therapies in colorectal cancer (CRC) patients. However, conflicting data exist regarding the variable response to EGFR-targeted therapy. The effects of oncogenic KRAS on downstream targets were studied in cell lines with different KRAS mutations. Cells harboring a single KRASG13D allele showed the most tumorigenic profile, with constitutive activation of the downstream pathway, rendering them EGF-unresponsive. Conversely, KRASA146T cells showed a full EGF-response in terms of signal transduction pathways, cell proliferation, migration or adhesion. Moreover, the global acetylome of CRC cells was also dependent on KRAS mutational status. Several hnRNP family members were identified within the 36 acetylated-proteins. Acetylation status is known to be involved in the modulation of EGF-response. In agreement with results presented herein, hnRNPA1 and L acetylation was induced in response to EGF in KRASA146T cells, whereas acetyl-hnRNPA1 and L levels remained unchanged after growth factor treatment in KRASG13D unresponsive cells. Our results showed that hnRNPs induced-acetylation is dependent on KRAS mutational status. Nevertheless hnRNPs acetylation might also be the point where different oncogenic pathways converge. | | 26110767
 |
MicroRNA-5p and -3p co-expression and cross-targeting in colon cancer cells.
Choo, KB; Soon, YL; Nguyen, PN; Hiew, MS; Huang, CJ
Journal of biomedical science
21
95
2014
Abstract anzeigen
Two mature miRNA species may be generated from the 5' and 3' arms of a pre-miRNA precursor. In most cases, only one species remains while the complementary species is degraded. However, co-existence of miRNA-5p and -3p species is increasingly being reported. In this work, we aimed to systematically investigate co-expression of miRNA-5p/3p in colon cancer cells in a genome-wide analysis, and to examine cross-targeting of the dysregulated miRNAs and 5p/3p species.Four colon cancer cell lines were examined relative to two normal colon tissues. Of the 1,190 miRNAs analyzed, 92 and 36 were found to be up- or down-regulated, respectively, in cancer cells. Nineteen co-expressed miRNA-5p/3p pairs were further identified suggesting frequent 5p/3p co-accumulation in colon cancer cells. Of these, 14 pairs were co-up-regulated and 3 pairs were co-down-regulated indicating concerted 5p/3p dysregulation. Nine dysregulated miRNA pairs fell into three miRNA gene families, namely let-7, mir-8/200 and mir-17, which showed frequent cross-targeting in the metastasis process. Focusing on the let-7d-5p/3p pair, the respectively targeted IGF1R and KRAS were shown to be in a reverse relationship with expression of the respective miRNA, which was confirmed in transient transfection assays using let-7d mimic or inhibitor. Targeting of KRAS by let-7d was previous reported; targeting of IGF1R by let-7d-5p was confirmed in luciferase assays in this study. The findings of let-7d-5p/3p and multiple other miRNAs targeting IGF1R, KRAS and other metastasis-related factors suggest that 5p/3p miRNAs contribute to cross-targeting of multiple cancer-associated factors and processes possibly to evade functional abolishment when any one of the crucial factors are inactivated.miRNA-5p/3p species are frequently co-expressed and are coordinately regulated in colon cancer cells. In cancer cells, multiple cross-targeting by the miRNAs, including the co-existing 5p/3p species, frequently occurs in an apparent safe-proof scheme of miRNA regulation of important tumorigenesis processes. Further systematic analysis of co-existing miRNA-5p/3p pairs in clinical tissues is important in elucidating 5p/3p contributions to cancer pathogenesis. | Western Blotting | 25287248
 |
MEK-dependent negative feedback underlies BCR-ABL-mediated oncogene addiction.
Asmussen, J; Lasater, EA; Tajon, C; Oses-Prieto, J; Jun, YW; Taylor, BS; Burlingame, A; Craik, CS; Shah, NP
Cancer discovery
4
200-15
2014
Abstract anzeigen
The clinical experience with BCR-ABL tyrosine kinase inhibitors (TKI) for the treatment of chronic myelogenous leukemia (CML) provides compelling evidence for oncogene addiction. Yet, the molecular basis of oncogene addiction remains elusive. Through unbiased quantitative phosphoproteomic analyses of CML cells transiently exposed to BCR-ABL TKI, we identified persistent downregulation of growth factor receptor (GF-R) signaling pathways. We then established and validated a tissue-relevant isogenic model of BCR-ABL-mediated addiction, and found evidence for myeloid GF-R signaling pathway rewiring that profoundly and persistently dampens physiologic pathway activation. We demonstrate that eventual restoration of ligand-mediated GF-R pathway activation is insufficient to fully rescue cells from a competing apoptotic fate. In contrast to previous work with BRAF(V600E) in melanoma cells, feedback inhibition following BCR-ABL TKI treatment is markedly prolonged, extending beyond the time required to initiate apoptosis. Mechanistically, BCR-ABL-mediated oncogene addiction is facilitated by persistent high levels of MAP-ERK kinase (MEK)-dependent negative feedback.We found that BCR–ABL can confer addiction in vitro by rewiring myeloid GF-R signaling through establishment of MEK-dependent negative feedback. Our findings predict that deeper, more durable responses to targeted agents across a range of malignancies may be facilitated by maintaining negative feedback concurrently with oncoprotein inhibition. | | 24362263
 |
A high-fat diet activates oncogenic Kras and COX2 to induce development of pancreatic ductal adenocarcinoma in mice.
Philip, B; Roland, CL; Daniluk, J; Liu, Y; Chatterjee, D; Gomez, SB; Ji, B; Huang, H; Wang, H; Fleming, JB; Logsdon, CD; Cruz-Monserrate, Z
Gastroenterology
145
1449-58
2013
Abstract anzeigen
Obesity is a risk factor for pancreatic ductal adenocarcinoma (PDAC), but it is not clear how obesity contributes to pancreatic carcinogenesis. The oncogenic form of KRAS is expressed during early stages of PDAC development and is detected in almost all of these tumors. However, there is evidence that mutant KRAS requires an additional stimulus to activate its full oncogenic activity and that this stimulus involves the inflammatory response. We investigated whether the inflammation induced by a high-fat diet, and the accompanying up-regulation of cyclooxygenase-2 (COX2), increases Kras activity during pancreatic carcinogenesis in mice.We studied mice with acinar cell-specific expression of KrasG12D (LSL-Kras/Ela-CreERT mice) alone or crossed with COX2 conditional knockout mice (COXKO/LSL-Kras/Ela-CreERT). We also studied LSL-Kras/PDX1-Cre mice. All mice were fed isocaloric diets with different amounts of fat, and a COX2 inhibitor was administered to some LSL-Kras/Ela-CreERT mice. Pancreata were collected from mice and analyzed for Kras activity, levels of phosphorylated extracellular-regulated kinase, inflammation, fibrosis, pancreatic intraepithelial neoplasia (PanIN), and PDACs.Pancreatic tissues from LSL-Kras/Ela-CreERT mice fed high-fat diets (HFDs) had increased Kras activity, fibrotic stroma, and numbers of PanINs and PDACs than LSL-Kras/Ela-CreERT mice fed control diets; the mice fed the HFDs also had shorter survival times than mice fed control diets. Administration of a COX2 inhibitor to LSL-Kras/Ela-CreERT mice prevented these effects of HFDs. We also observed a significant reduction in survival times of mice fed HFDs. COXKO/LSL-Kras/Ela-CreERT mice fed HFDs had no evidence for increased numbers of PanIN lesions, inflammation, or fibrosis, as opposed to the increases observed in LSL-Kras/Ela-CreERT mice fed HFDs.In mice, an HFD can activate oncogenic Kras via COX2, leading to pancreatic inflammation and fibrosis and development of PanINs and PDAC. This mechanism might be involved in the association between risk for PDAC and HFDs. | Western Blotting | 23958541
 |
Identification of mutant K-Ras-dependent phenotypes using a panel of isogenic cell lines.
Vartanian, S; Bentley, C; Brauer, MJ; Li, L; Shirasawa, S; Sasazuki, T; Kim, JS; Haverty, P; Stawiski, E; Modrusan, Z; Waldman, T; Stokoe, D
The Journal of biological chemistry
288
2403-13
2013
Abstract anzeigen
To assess the consequences of endogenous mutant K-Ras, we analyzed the signaling and biological properties of a small panel of isogenic cell lines. These include the cancer cell lines DLD1, HCT116, and Hec1A, in which either the WT or mutant K-ras allele has been disrupted, and SW48 colorectal cancer cells and human mammary epithelial cells in which a single copy of mutant K-ras was introduced at its endogenous genomic locus. We find that single copy mutant K-Ras causes surprisingly modest activation of downstream signaling to ERK and Akt. In contrast, a negative feedback signaling loop to EGFR and N-Ras occurs in some, but not all, of these cell lines. Mutant K-Ras also had relatively minor effects on cell proliferation and cell migration but more dramatic effects on cell transformation as assessed by growth in soft agar. Surprisingly, knock-out of the wild type K-ras allele consistently increased growth in soft agar, suggesting tumor-suppressive properties of this gene under these conditions. Finally, we examined the effects of single copy mutant K-Ras on global gene expression. Although transcriptional programs triggered by mutant K-Ras were generally quite distinct in the different cell lines, there was a small number of genes that were consistently overexpressed, and these could be used to monitor K-Ras inhibition in a panel of human tumor cell lines. We conclude that there are conserved components of mutant K-Ras signaling and phenotypes but that many depend on cell context and environmental cues. | | 23188824
 |
A phospholipase C-γ1-independent, RasGRP1-ERK-dependent pathway drives lymphoproliferative disease in linker for activation of T cells-Y136F mutant mice.
Kortum, RL; Rouquette-Jazdanian, AK; Miyaji, M; Merrill, RK; Markegard, E; Pinski, JM; Wesselink, A; Nath, NN; Alexander, CP; Li, W; Kedei, N; Roose, JP; Blumberg, PM; Samelson, LE; Sommers, CL
Journal of immunology (Baltimore, Md. : 1950)
190
147-58
2013
Abstract anzeigen
Mice expressing a germline mutation in the phospholipase C-γ1-binding site of linker for activation of T cells (LAT) show progressive lymphoproliferation and ultimately die at 4-6 mo age. The hyperactivated T cells in these mice show defective TCR-induced calcium flux but enhanced Ras/ERK activation, which is critical for disease progression. Despite the loss of LAT-dependent phospholipase C-γ1 binding and activation, genetic analysis revealed RasGRP1, and not Sos1 or Sos2, to be the major Ras guanine exchange factor responsible for ERK activation and the lymphoproliferative phenotype in these mice. Analysis of isolated CD4(+) T cells from LAT-Y136F mice showed altered proximal TCR-dependent kinase signaling, which activated a Zap70- and LAT-independent pathway. Moreover, LAT-Y136F T cells showed ERK activation that was dependent on Lck and/or Fyn, protein kinase C-θ, and RasGRP1. These data demonstrate a novel route to Ras activation in vivo in a pathological setting. | | 23209318
 |
Endomembrane H-Ras controls vascular endothelial growth factor-induced nitric-oxide synthase-mediated endothelial cell migration.
Haeussler, DJ; Pimentel, DR; Hou, X; Burgoyne, JR; Cohen, RA; Bachschmid, MM
The Journal of biological chemistry
288
15380-9
2013
Abstract anzeigen
We demonstrate for the first time that endomembrane-delimited H-Ras mediates VEGF-induced activation of endothelial nitric-oxide synthase (eNOS) and migratory response of human endothelial cells. Using thiol labeling strategies and immunofluorescent cell staining, we found that only 31% of total H-Ras is S-palmitoylated, tethering the small GTPase to the plasma membrane but leaving the function of the large majority of endomembrane-localized H-Ras unexplained. Knockdown of H-Ras blocked VEGF-induced PI3K-dependent Akt (Ser-473) and eNOS (Ser-1177) phosphorylation and nitric oxide-dependent cell migration, demonstrating the essential role of H-Ras. Activation of endogenous H-Ras led to recruitment and phosphorylation of eNOS at endomembranes. The loss of migratory response in cells lacking endogenous H-Ras was fully restored by modest overexpression of an endomembrane-delimited H-Ras palmitoylation mutant. These studies define a newly recognized role for endomembrane-localized H-Ras in mediating nitric oxide-dependent proangiogenic signaling. | Western Blotting | 23548900
 |
PLC-γ and PI3K link cytokines to ERK activation in hematopoietic cells with normal and oncogenic Kras.
Diaz-Flores, E; Goldschmidt, H; Depeille, P; Ng, V; Akutagawa, J; Krisman, K; Crone, M; Burgess, MR; Williams, O; Houseman, B; Shokat, K; Sampath, D; Bollag, G; Roose, JP; Braun, BS; Shannon, K
Science signaling
6
ra105
2013
Abstract anzeigen
Oncogenic K-Ras proteins, such as K-Ras(G12D), accumulate in the active, guanosine triphosphate (GTP)-bound conformation and stimulate signaling through effector kinases. The presence of the K-Ras(G12D) oncoprotein at a similar abundance to that of endogenous wild-type K-Ras results in only minimal phosphorylation and activation of the canonical Raf-mitogen-activated or extracellular signal-regulated protein kinase kinase (MEK)-extracellular signal-regulated kinase (ERK) and phosphoinositide 3-kinase (PI3K)-Akt-mammalian target of rapamycin (mTOR) signaling cascades in primary hematopoietic cells, and these pathways remain dependent on growth factors for efficient activation. We showed that phospholipase C-γ (PLC-γ), PI3K, and their generated second messengers link activated cytokine receptors to Ras and ERK signaling in differentiated bone marrow cells and in a cell population enriched for leukemia stem cells. Cells expressing endogenous oncogenic K-Ras(G12D) remained dependent on the second messenger diacylglycerol for the efficient activation of Ras-ERK signaling. These data raise the unexpected possibility of therapeutically targeting proteins that function upstream of oncogenic Ras in cancer. | | 24300897
 |
The RasGAP gene, RASAL2, is a tumor and metastasis suppressor.
McLaughlin, SK; Olsen, SN; Dake, B; De Raedt, T; Lim, E; Bronson, RT; Beroukhim, R; Polyak, K; Brown, M; Kuperwasser, C; Cichowski, K
Cancer cell
24
365-78
2013
Abstract anzeigen
RAS genes are commonly mutated in cancer; however, RAS mutations are rare in breast cancer, despite frequent hyperactivation of Ras and ERK. Here, we report that the RasGAP gene, RASAL2, functions as a tumor and metastasis suppressor. RASAL2 is mutated or suppressed in human breast cancer, and RASAL2 ablation promotes tumor growth, progression, and metastasis in mouse models. In human breast cancer, RASAL2 loss is associated with metastatic disease; low RASAL2 levels correlate with recurrence of luminal B tumors; and RASAL2 ablation promotes metastasis of luminal mouse tumors. Additional data reveal a broader role for RASAL2 inactivation in other tumor types. These studies highlight the expanding role of RasGAPs and reveal an alternative mechanism of activating Ras in cancer. | | 24029233
 |