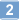Sirt1-deficiency causes defective protein quality control.
Tomita, T; Hamazaki, J; Hirayama, S; McBurney, MW; Yashiroda, H; Murata, S
Scientific reports
5
12613
2015
요약 표시
Protein quality control is an important mechanism to maintain cellular homeostasis. Damaged proteins have to be restored or eliminated by degradation, which is mainly achieved by molecular chaperones and the ubiquitin-proteasome system. The NAD(+)-dependent deacetylase Sirt1 has been reported to play positive roles in the regulation of cellular homeostasis in response to various stresses. However, its contribution to protein quality control remains unexplored. Here we show that Sirt1 is involved in protein quality control in both an Hsp70-dependent and an Hsp70-independent manner. Loss of Sirt1 led to the accumulation of ubiquitinated proteins in cells and tissues, especially upon heat stress, without affecting proteasome activities. This was partly due to decreased basal expression of Hsp70. However, this accumulation was only partially alleviated by overexpression of Hsp70 or induction of Hsp70 upon heat shock in Sirt1-deficient cells and tissues. These results suggest that Sirt1 mediates both Hsp70-dependent and Hsp70-independent protein quality control. Our findings cast new light on understanding the role of Sirt1 in maintaining cellular homeostasis. | | 26219988
 |
Endocytic Adaptor Protein Tollip Inhibits Canonical Wnt Signaling.
Toruń, A; Szymańska, E; Castanon, I; Wolińska-Nizioł, L; Bartosik, A; Jastrzębski, K; Miętkowska, M; González-Gaitán, M; Miaczynska, M
PloS one
10
e0130818
2015
요약 표시
Many adaptor proteins involved in endocytic cargo transport exhibit additional functions in other cellular processes which may be either related to or independent from their trafficking roles. The endosomal adaptor protein Tollip is an example of such a multitasking regulator, as it participates in trafficking and endosomal sorting of receptors, but also in interleukin/Toll/NF-κB signaling, bacterial entry, autophagic clearance of protein aggregates and regulation of sumoylation. Here we describe another role of Tollip in intracellular signaling. By performing a targeted RNAi screen of soluble endocytic proteins for their additional functions in canonical Wnt signaling, we identified Tollip as a potential negative regulator of this pathway in human cells. Depletion of Tollip potentiates the activity of β-catenin/TCF-dependent transcriptional reporter, while its overproduction inhibits the reporter activity and expression of Wnt target genes. These effects are independent of dynamin-mediated endocytosis, but require the ubiquitin-binding CUE domain of Tollip. In Wnt-stimulated cells, Tollip counteracts the activation of β-catenin and its nuclear accumulation, without affecting its total levels. Additionally, under conditions of ligand-independent signaling, Tollip inhibits the pathway after the stage of β-catenin stabilization, as observed in human cancer cell lines, characterized by constitutive β-catenin activity. Finally, the regulation of Wnt signaling by Tollip occurs also during early embryonic development of zebrafish. In summary, our data identify a novel function of Tollip in regulating the canonical Wnt pathway which is evolutionarily conserved between fish and humans. Tollip-mediated inhibition of Wnt signaling may contribute not only to embryonic development, but also to carcinogenesis. Mechanistically, Tollip can potentially coordinate multiple cellular pathways of trafficking and signaling, possibly by exploiting its ability to interact with ubiquitin and the sumoylation machinery. | | 26110841
 |
Coupled local translation and degradation regulate growth cone collapse.
Deglincerti, A; Liu, Y; Colak, D; Hengst, U; Xu, G; Jaffrey, SR
Nature communications
6
6888
2015
요약 표시
Local translation mediates axonal responses to Semaphorin3A (Sema3A) and other guidance cues. However, only a subset of the axonal proteome is locally synthesized, whereas most proteins are trafficked from the soma. The reason why only specific proteins are locally synthesized is unknown. Here we show that local protein synthesis and degradation are linked events in growth cones. We find that growth cones exhibit high levels of ubiquitination and that local signalling pathways trigger the ubiquitination and degradation of RhoA, a mediator of Sema3A-induced growth cone collapse. Inhibition of RhoA degradation is sufficient to remove the protein-synthesis requirement for Sema3A-induced growth cone collapse. In addition to RhoA, we find that locally translated proteins are the main targets of the ubiquitin-proteasome system in growth cones. Thus, local protein degradation is a major feature of growth cones and creates a requirement for local translation to replenish proteins needed to maintain growth cone responses. | | 25901863
 |
K63 polyubiquitination is a new modulator of the oxidative stress response.
Silva, GM; Finley, D; Vogel, C
Nature structural & molecular biology
22
116-23
2015
요약 표시
Ubiquitination is a post-translational modification that signals multiple processes, including protein degradation, trafficking and DNA repair. Polyubiquitin accumulates globally during the oxidative stress response, and this has been mainly attributed to increased ubiquitin conjugation and perturbations in protein degradation. Here we show that the unconventional Lys63 (K63)-linked polyubiquitin accumulates in the yeast Saccharomyces cerevisiae in a highly sensitive and regulated manner as a result of exposure to peroxides. We demonstrate that hydrogen peroxide inhibits the deubiquitinating enzyme Ubp2, leading to accumulation of K63 conjugates assembled by the Rad6 ubiquitin conjugase and the Bre1 ubiquitin ligase. Using linkage-specific isolation methods and stable isotope labeling by amino acids in cell culture (SILAC)-based quantitative proteomics, we identified greater than 100 new K63-polyubiquitinated targets, which were substantially enriched in ribosomal proteins. Finally, we demonstrate that impairment of K63 ubiquitination during oxidative stress affects polysome stability and protein expression, rendering cells more sensitive to stress, and thereby reveal a new redox-regulatory role for this modification. | | 25622294
 |
TRAF1 Coordinates Polyubiquitin Signaling to Enhance Epstein-Barr Virus LMP1-Mediated Growth and Survival Pathway Activation.
Greenfeld, H; Takasaki, K; Walsh, MJ; Ersing, I; Bernhardt, K; Ma, Y; Fu, B; Ashbaugh, CW; Cabo, J; Mollo, SB; Zhou, H; Li, S; Gewurz, BE
PLoS pathogens
11
e1004890
2015
요약 표시
The Epstein-Barr virus (EBV) encoded oncoprotein Latent Membrane Protein 1 (LMP1) signals through two C-terminal tail domains to drive cell growth, survival and transformation. The LMP1 membrane-proximal TES1/CTAR1 domain recruits TRAFs to activate MAP kinase, non-canonical and canonical NF-kB pathways, and is critical for EBV-mediated B-cell transformation. TRAF1 is amongst the most highly TES1-induced target genes and is abundantly expressed in EBV-associated lymphoproliferative disorders. We found that TRAF1 expression enhanced LMP1 TES1 domain-mediated activation of the p38, JNK, ERK and canonical NF-kB pathways, but not non-canonical NF-kB pathway activity. To gain insights into how TRAF1 amplifies LMP1 TES1 MAP kinase and canonical NF-kB pathways, we performed proteomic analysis of TRAF1 complexes immuno-purified from cells uninduced or induced for LMP1 TES1 signaling. Unexpectedly, we found that LMP1 TES1 domain signaling induced an association between TRAF1 and the linear ubiquitin chain assembly complex (LUBAC), and stimulated linear (M1)-linked polyubiquitin chain attachment to TRAF1 complexes. LMP1 or TRAF1 complexes isolated from EBV-transformed lymphoblastoid B cell lines (LCLs) were highly modified by M1-linked polyubiqutin chains. The M1-ubiquitin binding proteins IKK-gamma/NEMO, A20 and ABIN1 each associate with TRAF1 in cells that express LMP1. TRAF2, but not the cIAP1 or cIAP2 ubiquitin ligases, plays a key role in LUBAC recruitment and M1-chain attachment to TRAF1 complexes, implicating the TRAF1:TRAF2 heterotrimer in LMP1 TES1-dependent LUBAC activation. Depletion of either TRAF1, or the LUBAC ubiquitin E3 ligase subunit HOIP, markedly impaired LCL growth. Likewise, LMP1 or TRAF1 complexes purified from LCLs were decorated by lysine 63 (K63)-linked polyubiqutin chains. LMP1 TES1 signaling induced K63-polyubiquitin chain attachment to TRAF1 complexes, and TRAF2 was identified as K63-Ub chain target. Co-localization of M1- and K63-linked polyubiquitin chains on LMP1 complexes may facilitate downstream canonical NF-kB pathway activation. Our results highlight LUBAC as a novel potential therapeutic target in EBV-associated lymphoproliferative disorders. | Western Blotting | 25996949
 |
Cellular inhibitor of apoptosis (cIAP)-mediated ubiquitination of phosphofurin acidic cluster sorting protein 2 (PACS-2) negatively regulates tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) cytotoxicity.
Guicciardi, ME; Werneburg, NW; Bronk, SF; Franke, A; Yagita, H; Thomas, G; Gores, GJ
PloS one
9
e92124
2014
요약 표시
Lysosomal membrane permeabilization is an essential step in TRAIL-induced apoptosis of liver cancer cell lines. TRAIL-induced lysosomal membrane permeabilization is mediated by the multifunctional sorting protein PACS-2 and repressed by the E3 ligases cIAP-1 and cIAP-2. Despite the opposing roles for PACS-2 and cIAPs in TRAIL-induced apoptosis, an interaction between these proteins has yet to be examined. Herein, we report that cIAP-1 and cIAP-2 confer TRAIL resistance to hepatobiliary cancer cell lines by reducing PACS-2 levels. Under basal conditions, PACS-2 underwent K48-linked poly-ubiquitination, resulting in PACS-2 proteasomal degradation. Biochemical assays showed cIAP-1 and cIAP-2 interacted with PACS-2 in vitro and co-immunoprecipitation studies demonstrated that the two cIAPs bound PACS-2 in vivo. More importantly, both cIAP-1 and cIAP-2 directly mediated PACS-2 ubiquitination in a cell-free assay. Single c-Iap-1 or c-Iap-2 gene knock-outs in mouse hepatocytes did not lead to PACS-2 accumulation. However, deletion of both cIAP-1 and cIAP-2 reduced PACS-2 ubiquitination, which increased PACS-2 levels and sensitized HuH-7 cells to TRAIL-induced lysosomal membrane permeabilization and apoptosis. Correspondingly, deletion of cIAPs sensitized wild-type, but not PACS-2-deficient hepatocarcinoma cells or Pacs-2-/- mouse hepatocytes to TRAIL-induced apoptosis. Together, these data suggest cIAPs constitutively downregulate PACS-2 by polyubiquitination and proteasomal degradation, thereby restraining TRAIL-induced killing of liver cancer cells. | | 24633224
 |
Differential roles of the ubiquitin proteasome system and autophagy in the clearance of soluble and aggregated TDP-43 species.
Scotter, EL; Vance, C; Nishimura, AL; Lee, YB; Chen, HJ; Urwin, H; Sardone, V; Mitchell, JC; Rogelj, B; Rubinsztein, DC; Shaw, CE
Journal of cell science
127
1263-78
2014
요약 표시
TAR DNA-binding protein (TDP-43, also known as TARDBP) is the major pathological protein in amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). Large TDP-43 aggregates that are decorated with degradation adaptor proteins are seen in the cytoplasm of remaining neurons in ALS and FTD patients post mortem. TDP-43 accumulation and ALS-linked mutations within degradation pathways implicate failed TDP-43 clearance as a primary disease mechanism. Here, we report the differing roles of the ubiquitin proteasome system (UPS) and autophagy in the clearance of TDP-43. We have investigated the effects of inhibitors of the UPS and autophagy on the degradation, localisation and mobility of soluble and insoluble TDP-43. We find that soluble TDP-43 is degraded primarily by the UPS, whereas the clearance of aggregated TDP-43 requires autophagy. Cellular macroaggregates, which recapitulate many of the pathological features of the aggregates in patients, are reversible when both the UPS and autophagy are functional. Their clearance involves the autophagic removal of oligomeric TDP-43. We speculate that, in addition to an age-related decline in pathway activity, a second hit in either the UPS or the autophagy pathway drives the accumulation of TDP-43 in ALS and FTD. Therapies for clearing excess TDP-43 should therefore target a combination of these pathways. | | 24424030
 |
Cell fate decisions regulated by K63 ubiquitination of tumor necrosis factor receptor 1.
Fritsch, J; Stephan, M; Tchikov, V; Winoto-Morbach, S; Gubkina, S; Kabelitz, D; Schütze, S
Molecular and cellular biology
34
3214-28
2014
요약 표시
Signaling by tumor necrosis factor (TNF) receptor 1 (TNF-R1), a prototypic member of the death receptor family, mediates pleiotropic biological outcomes ranging from inflammation and cell proliferation to cell death. Although many elements of specific signaling pathways have been identified, the main question of how these selective cell fate decisions are regulated is still unresolved. Here we identified TNF-induced K63 ubiquitination of TNF-R1 mediated by the ubiquitin ligase RNF8 as an early molecular checkpoint in the regulation of the decision between cell death and survival. Downmodulation of RNF8 prevented the ubiquitination of TNF-R1, blocked the internalization of the receptor, prevented the recruitment of the death-inducing signaling complex and the activation of caspase-8 and caspase-3/7, and reduced apoptotic cell death. Conversely, recruitment of the adaptor proteins TRADD, TRAF2, and RIP1 to TNF-R1, as well as activation of NF-κB, was unimpeded and cell growth and proliferation were significantly enhanced in RNF8-deficient cells. Thus, K63 ubiquitination of TNF-R1 can be sensed as a new level of regulation of TNF-R1 signaling at the earliest stage after ligand binding. | | 24980434
 |
Intramitochondrial recruitment of endolysosomes mediates Smac degradation and constitutes a novel intrinsic apoptosis antagonizing function of XIAP E3 ligase.
Hamacher-Brady, A; Choe, SC; Krijnse-Locker, J; Brady, NR
Cell death and differentiation
21
1862-76
2014
요약 표시
Intrinsic apoptosis involves BH3-only protein activation of Bax/Bak-mediated mitochondrial outer membrane permeabilization (MOMP). Consequently, cytochrome c is released from the mitochondria to activate caspases, and Smac (second mitochondria-derived activator of caspases) to inhibit XIAP-mediated caspase suppression. Dysfunctional mitochondria can be targeted for lysosomal degradation via autophagy (mitophagy), or directly through mitochondria-derived vesicle transport. However, the extent of autophagy and lysosomal interactions with apoptotic mitochondria remains largely unknown. We describe here a novel pathway of endolysosomal processing of mitochondria, activated in response to canonical BH3-only proteins and mitochondrial depolarization. We report that expression of canonical BH3-only proteins, tBid, BimEL, Bik, Bad, and mitophagy receptor mutants of atypical BH3-only proteins, Bnip3 and Bnip3L/Nix, leads to prominent relocalization of endolysosomes into inner mitochondrial compartments, in a manner independent of mitophagy. As an upstream regulator, we identified the XIAP E3 ligase. In response to mitochondrial depolarization, XIAP actuates Bax-mediated MOMP, even in the absence of BH3-only protein signaling. Subsequently, in an E3 ligase-dependent manner, XIAP rapidly localizes inside all the mitochondria, and XIAP-mediated mitochondrial ubiquitylation catalyses interactions of Rab membrane targeting components Rabex-5 and Rep-1 (RFP-tagged Rab escort protein-1), and Rab5- and Rab7-positive endolysosomes, at and within mitochondrial membrane compartments. While XIAP-mediated MOMP permits delayed cytochrome c release, within the mitochondria XIAP selectively signals lysosome- and proteasome-associated degradation of its inhibitor Smac. These findings suggest a general mechanism to lower the mitochondrial apoptotic potential via intramitochondrial degradation of Smac. | | 25080938
 |
Toll-like receptor alterations in myelodysplastic syndrome.
Wei, Y; Dimicoli, S; Bueso-Ramos, C; Chen, R; Yang, H; Neuberg, D; Pierce, S; Jia, Y; Zheng, H; Wang, H; Wang, X; Nguyen, M; Wang, SA; Ebert, B; Bejar, R; Levine, R; Abdel-Wahab, O; Kleppe, M; Ganan-Gomez, I; Kantarjian, H; Garcia-Manero, G
Leukemia
27
1832-40
2013
요약 표시
Recent studies have implicated the innate immunity system in the pathogenesis of myelodysplastic syndromes (MDS). Toll-like receptor (TLR) genes encode key innate immunity signal initiators. We recently identified multiple genes, known to be regulated by TLRs, to be overexpressed in MDS bone marrow (BM) CD34+ cells, and hypothesized that TLR signaling is abnormally activated in MDS. We analyzed a large cohort of MDS cases and identified TLR1, TLR2 and TLR6 to be significantly overexpressed in MDS BM CD34+ cells. Deep sequencing followed by Sanger resequencing of TLR1, TLR2, TLR4 and TLR6 genes uncovered a recurrent genetic variant, TLR2-F217S, in 11% of 149 patients. Functionally, TLR2-F217S results in enhanced activation of downstream signaling including NF-κB activity after TLR2 agonist treatment. In cultured primary BM CD34+ cells of normal donors, TLR2 agonists induced histone demethylase JMJD3 and interleukin-8 gene expression. Inhibition of TLR2 in BM CD34+ cells from patients with lower-risk MDS using short hairpin RNA resulted in increased erythroid colony formation. Finally, RNA expression levels of TLR2 and TLR6, as well as presence of TLR2-F217S, are associated with distinct prognosis and clinical characteristics. These findings indicate that TLR2-centered signaling is deregulated in MDS, and that its targeting may have potential therapeutic benefit in MDS. | | 23765228
 |