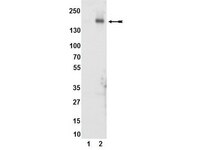
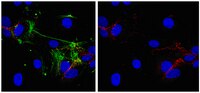
Phosphorylation of Akt and ERK1/2 is required for VEGF-A/VEGFR2-induced proliferation and migration of lymphatic endothelium.
Dellinger, MT; Brekken, RA
PloS one
6
e28947
2011
概要を表示する
There is growing evidence that vascular endothelial growth factor-A (VEGF-A), a ligand of the receptor tyrosine kinases VEGFR1 and VEGFR2, promotes lymphangiogenesis. However, the underlying mechanisms by which VEGF-A induces the growth of lymphatic vessels remain poorly defined. Here we report that VEGFR2, not VEGFR1, is the primary receptor regulating VEGF-A-induced lymphangiogenesis. We show that specific inhibition of VEGF-A/VEGFR2 signaling with the fully human monoclonal antibody r84 significantly inhibits lymphangiogenesis in MDA-MB-231 tumors. In vitro experiments with primary human dermal lymphatic endothelial cells (LECs) demonstrate that blocking VEGF-A activation of VEGFR2, not VEGFR1, significantly inhibits VEGF-A-induced proliferation and migration of LECs. We show that VEGF-A stimulation of LECs leads to the phosphorylation of VEGFR2 (Tyr 951, 1054, 1059, 1175, and 1214) which subsequently triggers PKC dependent phosphorylation of ERK1/2 and PI3-K dependent phosphorylation of Akt. Additionally, we demonstrate that inhibitors that suppress the phosphorylation of ERK1/2 and Akt significantly block VEGF-A- induced proliferation and migration of LECs. Together, these results shed light on the mechanisms regulating VEGF-A-induced proliferation and migration of LECs, reveal that VEGFR2 is the primary signaling VEGF-A receptor on lymphatic endothelium, and suggest that therapeutic agents targeting the VEGF-A/VEGFR2 axis could be useful in blocking the pathological formation of lymphatic vessels. 記事全文 | 22174934
 |