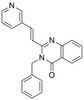
The heparan sulfate proteoglycan form of epithelial CD44v3 serves as a CD11b/CD18 counter-receptor during polymorphonuclear leukocyte transepithelial migration.
Zen, K; Liu, DQ; Li, LM; Chen, CX; Guo, YL; Ha, B; Chen, X; Zhang, CY; Liu, Y
The Journal of biological chemistry
284
3768-76
2009
概要を表示する
Leukocyte beta2-integrin CD11b/CD18 mediates the firm adhesion and subsequent transepithelial migration of polymorphonuclear leukocytes, but the identity of its counter-receptor(s) on epithelia remains elusive. Here we identified a monoclonal antibody, clone C3H7, which strongly bound to the basolateral membranes of epithelial cells and inhibited both the adhesion of epithelial cells to immobilized CD11b/CD8 and the transepithelial migration of PMNs in a physiologically relevant basolateral-to-apical direction. C3H7 antigen expression in epithelial monolayers was significantly increased by treatment with proinflammatory cytokine interferon-gamma or a combination of interferon-gamma and tumor necrosis factor-alpha. Up-regulation of C3H7 antigen was also observed in the epithelium of inflamed human colon tissues. Microsequencing and Western blotting of the purified antigen showed it to be CD44 variant 3 (CD44v3), a approximately 160-kDa membrane glycoprotein. Further studies demonstrated that this epithelial CD44v3 specifically binds to CD11b/CD18 through its heparan sulfate moieties. In summary, our study demonstrates for the first time that the heparan sulfate proteoglycan form of epithelial CD44v3 plays a critical role in facilitating PMN recruitment during inflammatory episodes via directly binding to CD11b/CD18. 記事全文 | | 19073595
 |
Syndecan-1/CD147 association is essential for cyclophilin B-induced activation of p44/42 mitogen-activated protein kinases and promotion of cell adhesion and chemotaxis.
Pakula, R; Melchior, A; Denys, A; Vanpouille, C; Mazurier, J; Allain, F
Glycobiology
17
492-503
2007
概要を表示する
Many of the biological functions attributed to cell surface proteoglycans are dependent on the interaction with extracellular mediators through their heparan sulphate (HS) moieties and the participation of their core proteins in signaling events. A class of recently identified inflammatory mediators is secreted cyclophilins, which are mostly known as cyclosporin A-binding proteins. We previously demonstrated that cyclophilin B (CyPB) triggers chemotaxis and integrin-mediated adhesion of T lymphocytes mainly of the CD4+/CD45RO+ phenotype. These activities are related to interactions with two types of binding sites, CD147 and cell surface HS. Here, we demonstrate that CyPB-mediated adhesion of CD4+/CD45RO+ T cells is related to p44/42 mitogen-activated protein kinase (MAPK) activation by a mechanism involving CD147 and HS proteoglycans (HSPG). Although HSPG core proteins are represented by syndecan-1, -2, -4, CD44v3 and betaglycan in CD4+/CD45RO+ T cells, we found that only syndecan-1 is physically associated with CD147. The intensity of the heterocomplex increased in response to CyPB, suggesting a transient enhancement and/or stabilization in the association of CD147 to syndecan-1. Pretreatment with anti-syndecan-1 antibodies or knockdown of syndecan-1 expression by RNA interference dramatically reduced CyPB-induced p44/p42 MAPK activation and consequent migration and adhesion, supporting the model in which syndecan-1 serves as a binding subunit to form the fully active receptor of CyPB. Altogether, our findings provide a novel example of a soluble mediator in which a member of the syndecan family plays a critical role in efficient interaction with signaling receptors and initiation of cellular responses. | | 17267519
 |
Surface protein expression and messenger RNA-splicing analysis of CD44 in uterine cervical cancer and normal cervical epithelium.
Dall, P; Heider, KH; Hekele, A; von Minckwitz, G; Kaufmann, M; Ponta, H; Herrlich, P
Cancer research
3337-41
1994
概要を表示する
Variant CD44 has recently been shown to serve as a metastasis marker in human breast cancer. Certain variant epitopes on primary tumors predict poor survival probabilities for the patients. In this study, immunohistochemical analysis of 16 uterine cervical carcinomas showed strong expression of several CD44 variant epitopes in all samples. In normal cervical epithelia from 5 patients, expression of these epitopes was restricted to particular cell layers, with expression being strong in basal and spinal cells but absent in superficial cells. Fifteen of 16 cancer samples were stained strongly with an antibody which recognizes one particular CD44 epitope that is encoded by both variant exons v7 and v8. This epitope was not detectable in normal cervical epithelium. CD44-mRNA splicing analysis showed qualitative and quantitative differences between malignant and normal tissues with a much more complex splice pattern and high expression of a large CD44 isoform containing variant exons v3 to v10 (including the v7/v8 transition epitope) in about one-half of the cancer samples. Interestingly, patients with lymph node metastases were in this group only. These differences in CD44 epitope expression and mRNA splicing in cervical carcinoma reveal dynamic changes in CD44 expression during carcinogenesis. Such changes could provide metastatic cells with a selective advantage during the carcinogenic process. Furthermore, the v7/v8 epitope may be suitable for screening early stages of cervical cancer. | Immunofluorescence | 7516819
 |
Genomic structure of DNA encoding the lymphocyte homing receptor CD44 reveals at least 12 alternatively spliced exons.
Screaton, G R, et al.
Proc. Natl. Acad. Sci. U.S.A., 89: 12160-4 (1992)
1992
概要を表示する
The CD44 molecule is known to display extensive size heterogeneity, which has been attributed both to alternative splicing and to differential glycosylation within the extracellular domain. Although the presence of several alternative exons has been partly inferred from cDNA sequencing, the precise intron-exon organization of the CD44 gene has not been described to date to our knowledge. In the present study we describe the structure of the human CD44 gene, which contains at least 19 exons spanning some 50 kilobases of DNA. We have identified 10 alternatively spliced exons within the extracellular domain, including 1 exon that has not been previously reported. In addition to the inclusion or exclusion of whole exons, more diversity is generated through the utilization of internal splice donor and acceptor sites within 2 of the individual exons. The variation previously reported for the cytoplasmic domain is shown to result from the alternative splicing of 2 exons. The genomic structure of CD44 reveals a remarkable degree of complexity, and we confirm the role of alternative splicing as the basis of the structural and functional diversity seen in the CD44 molecule. | | 1465456
 |
Participation in normal immune responses of a metastasis-inducing splice variant of CD44.
Arch, R, et al.
Science, 257: 682-5 (1992)
1992
概要を表示する
A variant of the glycoprotein CD44 (CD44v) that shares sequences with variants causally involved in metastasis formation is transiently expressed on B and T lymphocytes and macrophages after antigenic stimulation and in the postnatal period. Antibodies to the variant hinder in vivo activation of both B and T cells. The observation that a protein domain that is expressed on CD44 and required for the lymphatic spread of tumor cells can catalyze an essential step in the process of lymphocyte activation supports the idea that metastasizing tumor cells mimic lymphocyte behavior. | | 1496383
 |