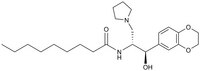
538285 Sigma-AldrichGlucosylceramide Synthase Inhibitor, Genz-123346 - CAS 491833-30-8 - Calbiochem
Sinónimos: (1R,2R)-nonanoic acid[2-(2ʹ,3ʹ-dihydro-benzo [1, 4] dioxin-6ʹ-yl)-2-hydroxy-1-pyrrolidin-1-ylmethyl-ethyl]-amide, GL1 synthase Inhibitor, Genz-123346, Genz123346
Productos recomendados
Descripción
| Replacement Information |
|---|
Tabla espec. clave
| CAS # | Empirical Formula |
|---|---|
| 491833-30-8 | C₂₄H₃₈N₂O₄ |
Precios y disponibilidad
| Número de referencia | Disponiblidad | Embalaje | Cant./Env. | Precio | Cantidad | |
|---|---|---|---|---|---|---|
| 5.38285.0001 |
|
Frasco de vidrio | 10 mg |
|
— |
| References | |
|---|---|
| References | Shen, W., et al. 2014. J. Neurochem. 129, 884. Natoli, T.A., et al. 2010. Nat. Med. 16, 788. Zhao, H., et al. 2007. Diabetes. 56, 1210. |
| Product Information | |
|---|---|
| CAS number | 491833-30-8 |
| Form | White to off-white solid |
| Hill Formula | C₂₄H₃₈N₂O₄ |
| Chemical formula | C₂₄H₃₈N₂O₄ |
| Reversible | Y |
| Quality Level | MQ100 |
| Applications |
|---|
| Biological Information | |
|---|---|
| Primary Target | glucosylceramide synthase |
| Primary Target IC<sub>50</sub> | 14 nM for ganglioside GM1 suppression |
| Purity | ≥98% by HPLC |
| Physicochemical Information | |
|---|---|
| Cell permeable | Y |
| Dimensions |
|---|
| Materials Information |
|---|
| Toxicological Information |
|---|
| Safety Information according to GHS |
|---|
| Safety Information |
|---|
| Product Usage Statements |
|---|
| Packaging Information | |
|---|---|
| Packaged under inert gas | Packaged under inert gas |
| Transport Information |
|---|
| Supplemental Information |
|---|
| Specifications |
|---|
| Global Trade Item Number | |
|---|---|
| Número de referencia | GTIN |
| 5.38285.0001 | 04054839119569 |
Documentation
Glucosylceramide Synthase Inhibitor, Genz-123346 - CAS 491833-30-8 - Calbiochem Ficha datos de seguridad (MSDS)
| Título |
|---|
Referencias bibliográficas
| Visión general referencias |
|---|
| Shen, W., et al. 2014. J. Neurochem. 129, 884. Natoli, T.A., et al. 2010. Nat. Med. 16, 788. Zhao, H., et al. 2007. Diabetes. 56, 1210. |