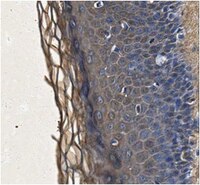
Expression and localization of the two small proteoglycans biglycan and decorin in developing human skeletal and non-skeletal tissues.
Bianco, P; Fisher, LW; Young, MF; Termine, JD; Robey, PG
The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society
38
1549-63
1990
Show Abstract
The messenger RNAs and core proteins of the two small chondroitin/dermatan sulfate proteoglycans, biglycan and decorin, were localized in developing human bone and other tissues by both 35S-labeled RNA probes and antibodies directed against synthetic peptides corresponding to nonhomologous regions of the two core proteins. Biglycan and decorin expression and localization were substantially divergent and sometimes mutually exclusive. In developing bones, spatially restricted patterns of gene expression and/or matrix localization of the two proteoglycans were identified in articular regions, epiphyseal cartilage, vascular canals, subperichondral regions, and periosteum, and indicated the association of each molecule with specific developmental events at specific sites. Study of non-skeletal tissues revealed that decorin was associated with all major type I (and type II) collagen-rich connective tissues. Conversely, biglycan was expressed and localized in a range of specialized cell types, including connective tissue (skeletal myofibers, endothelial cells) and epithelial cells (differentiating keratinocytes, renal tubular epithelia). Biglycan core protein was localized at the cell surface of certain cell types (e.g., keratinocytes). Whereas the distribution of decorin was consistent with matrix-centered functions, possibly related to regulation of growth of collagen fibers, the distribution of biglycan pointed to other function(s), perhaps related to cell regulation. | 2212616
 |
Deduced protein sequence of bone small proteoglycan I (biglycan) shows homology with proteoglycan II (decorin) and several nonconnective tissue proteins in a variety of species.
Fisher, LW; Termine, JD; Young, MF
The Journal of biological chemistry
264
4571-6
1989
Show Abstract
The small proteoglycans (PG) of bone consist of two different molecular species: one containing one chondroitin sulfate chain (PG II) and the other, two chains (PG I). These two proteoglycans are found in many connective tissues and have Mr = 45,000 core proteins with clear differences in their NH2-terminal sequences. Using antisera produced against synthetic peptides derived from the human PG I and PG II NH2 termini, we have isolated several cDNA clones from a lambda gt11 expression library made against mRNA isolated from human bone-derived cells. The clones, which reacted with antisera to the PG II peptide, were sequenced and found to be identical with the PG II class of proteoglycan from human fibroblasts known as PG-40 or decorin. The clones reacting to the PG I antisera, however, had a unique sequence. The derived protein sequence of PG I showed sufficient homology with the PG II sequence (55% of the amino acids are identical, with most others involving chemically similar amino acid substitutions) to strongly suggest that the two proteins were the result of a gene duplication. PG II (decorin) contains one attached glycosaminoglycan chain, while PG I probably contains two chains. For this reason, we suggest that PG I be called biglycan. The biglycan protein sequence contains 368 residues (Mr = 42,510 for the complete sequence and Mr = 37,983 for the secreted form) that appears to consist predominantly of a series of 12 tandem repeats of 24 residues. The repeats are recognized by their conserved leucines (and leucine-like amino acids) in positions previously reported for a diverse collection of proteins (none of which is thought to be proteoglycans) including: two morphogenic proteins (toll and chaoptin) in the fruit fly; a yeast adenylate cyclase; and two human proteins, the von Willebrand Factor-binding platelet membrane protein, GPIb, and a rare serum protein, leucine-rich glycoprotein. | 2647739
 |