533717 Sigma-AldrichGLS1 Inhibitor III, CB-839 - CAS 1439399-58-2 - Calbiochem
Cell-permeable, orally available, potent, selective & non-competitive GLS1 inhibitor. Reduces glutamine consumption & glutamate secretion, and induces apoptosis.
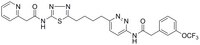
More>> Cell-permeable, orally available, potent, selective & non-competitive GLS1 inhibitor. Reduces glutamine consumption & glutamate secretion, and induces apoptosis. Less<<Synonyms: N-(6-(4-(5-((2-Pyridin-2-ylacetyl)amino)-1,3,4-thiadiazol-2-yl)butyl)pyridazin-3-yl)-2-(3-(trifluoromethoxy)phenyl)acetamide, KGA Inhibitor III, 2-(Pyridin-2-yl)-N-(5-(4-(6-(2-(3-(trifluoromethoxy)phenyl)acetamido)pyridazin-3-yl)butyl)-1,3,4-thiadiazol-2-yl)acetamide, GAC Inhibitor III, Kidney-Type Glutaminase Inhibitor III
Recommended Products
Overview
| Replacement Information |
|---|
Key Spec Table
| CAS # | Empirical Formula |
|---|---|
| 1439399-58-2 | C₂₆H₂₄F₃N₇O₃S |
Pricing & Availability
| Catalogue Number | Availability | Packaging | Qty/Pack | Price | Quantity | |
|---|---|---|---|---|---|---|
| 5.33717.0001 |
|
Botol kaca | 10 mg |
|
— |
| References | |
|---|---|
| References | Gross, M.I., et al. 2014. Mol. Cancer Ther. 13, 890. |
| Product Information | |
|---|---|
| CAS number | 1439399-58-2 |
| Form | Yellow to brown solid |
| Hill Formula | C₂₆H₂₄F₃N₇O₃S |
| Chemical formula | C₂₆H₂₄F₃N₇O₃S |
| Reversible | Y |
| Quality Level | MQ100 |
| Applications |
|---|
| Biological Information | |
|---|---|
| Primary Target | GLS1 |
| Primary Target IC<sub>50</sub> | 23 nM & 28 nM, respectively, using murine kidney and brain homogenates |
| Purity | ≥97% by HPLC |
| Physicochemical Information | |
|---|---|
| Cell permeable | Y |
| Dimensions |
|---|
| Materials Information |
|---|
| Toxicological Information |
|---|
| Safety Information according to GHS |
|---|
| Safety Information |
|---|
| Product Usage Statements |
|---|
| Packaging Information | |
|---|---|
| Packaged under inert gas | Packaged under inert gas |
| Transport Information |
|---|
| Supplemental Information |
|---|
| Specifications |
|---|
| Global Trade Item Number | |
|---|---|
| Catalogue Number | GTIN |
| 5.33717.0001 | 04055977286656 |
Documentation
GLS1 Inhibitor III, CB-839 - CAS 1439399-58-2 - Calbiochem MSDS
| Title |
|---|
GLS1 Inhibitor III, CB-839 - CAS 1439399-58-2 - Calbiochem Certificates of Analysis
| Title | Lot Number |
|---|---|
| 533717 |
References
| Reference overview |
|---|
| Gross, M.I., et al. 2014. Mol. Cancer Ther. 13, 890. |
| Data Sheet | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Note that this data sheet is not lot-specific and is representative of the current specifications for this product. Please consult the vial label and the certificate of analysis for information on specific lots. Also note that shipping conditions may differ from storage conditions.
|