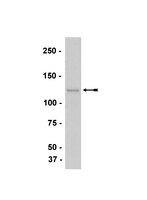
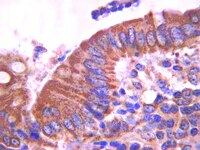
Convergence of Wnt, beta-catenin, and cadherin pathways.
Nelson, W James and Nusse, Roel
Science, 303: 1483-7 (2004)
2004
Kivonat megmutatása
The specification and proper arrangements of new cell types during tissue differentiation require the coordinated regulation of gene expression and precise interactions between neighboring cells. Of the many growth factors involved in these events, Wnts are particularly interesting regulators, because a key component of their signaling pathway, beta-catenin, also functions as a component of the cadherin complex, which controls cell-cell adhesion and influences cell migration. Here, we assemble evidence of possible interrelations between Wnt and other growth factor signaling, beta-catenin functions, and cadherin-mediated adhesion. | 15001769
 |
The cadherin-catenin complex as a focal point of cell adhesion and signalling: new insights from three-dimensional structures.
Gooding, Jane M, et al.
Bioessays, 26: 497-511 (2004)
2004
Kivonat megmutatása
Cadherins are a large family of single-pass transmembrane proteins principally involved in Ca2+-dependent homotypic cell adhesion. The cadherin molecules comprise three domains, the intracellular domain, the transmembrane domain and the extracellular domain, and form large complexes with a vast array of binding partners (including cadherin molecules of the same type in homophilic interactions and cellular protein catenins), orchestrating biologically essential extracellular and intracellular signalling processes. While current, contrasting models for classic cadherin homophilic interaction involve varying numbers of specific repeats found in the extracellular domain, the structure of the domain itself clearly remains the main determinant of cell stability and binding specificity. Through intracellular interactions, cadherin enhances its adhesive properties binding the cytoskeleton via cytoplasmic associated factors alpha- catenin, beta-catenin and p120ctn. Recent structural studies on classic cadherins and these catenin molecules have provided new insight into the essential mechanisms underlying cadherin-mediated cell interaction and catenin-mediated cellular signalling. Remarkable structural diversity has been observed in beta-catenin recognition of other cellular factors including APC, Tcf and ICAT, proteins that contribute to or compete with cadherin/catenin functioning. | 15112230
 |
Structure-based models of cadherin-mediated cell adhesion: the evolution continues.
Koch, A W, et al.
Cell. Mol. Life Sci., 61: 1884-95 (2004)
2004
Kivonat megmutatása
Cadherins are glycoproteins that are responsible for homophilic, Ca2+-dependent cell-cell adhesion and play crucial roles in many cellular adhesion processes ranging from embryogenesis to the formation of neuronal circuits in the central nervous system. Many different experimental approaches have been used to unravel the molecular basis for cadherin-mediated adhesion. In particular, several high-resolution structures have provided models for cadherin-cadherin interactions that are illuminative in many respects yet contradictory in others. This review gives an overview of the structural studies of cadherins over the past decade while focusing on recent developments that reconcile some of the earlier findings. | 15289931
 |
M-cadherin and its sisters in development of striated muscle.
Kaufmann, U, et al.
Cell Tissue Res., 296: 191-8 (1999)
1998
Kivonat megmutatása
Cadherins are calcium-dependent, transmembrane intercellular adhesion proteins with morphoregulatory functions in the development and maintenance of tissues. In the development of striated muscle, the expression and function of mainly M-, N-, and R-cadherin has been studied so far. While these three cadherins are expressed in skeletal muscle cells, of these only N-cadherin is expressed in cardiac muscle. In this review, M-, N-, and R-cadherin are discussed as important players in the terminal differentiation and possibly also in the commitment of skeletal muscle cells. Furthermore, reports are described which evaluate the essential role of N-cadherin in the formation of heart tissue. | 10199979
 |
Molecular cloning and characterization of a novel human classic cadherin homologous with mouse muscle cadherin.
Shimoyama, Y, et al.
J. Biol. Chem., 273: 10011-8 (1998)
1998
Kivonat megmutatása
We used a novel cDNA cloning method based on the cadherin-beta-catenin protein interaction and identified a new human classic-type cadherin, which we named cadherin-15, from adult brain and skeletal muscle cDNA libraries. Sequence analysis revealed that this cadherin was closely related to mouse muscle cadherin and seemed to be its human counterpart. However, its deduced amino acid sequence differed from that of mouse muscle cadherin in that it had an extra 31-amino acid sequence at its C terminus that has been found neither in mouse muscle cadherin nor in any other known classic cadherin. Analysis of cadherin-15 protein expressed in L fibroblasts showed that it was cleaved proteolytically, expressed on the cell surfaces as a mature form of about 124-kDa, and functioned as a cell-cell adhesion molecule in a homophilic and specific manner, but Ca2+ did not protect it against degradation by trypsin. Our findings also suggest that cadherin-15 mediates cell-cell adhesion with a binding strength comparable to that of E-cadherin. | 9545347
 |