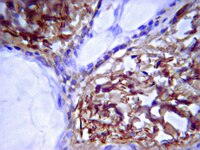
Mural cell associated VEGF is required for organotypic vessel formation.
Evensen, L; Micklem, DR; Blois, A; Berge, SV; Aarsaether, N; Littlewood-Evans, A; Wood, J; Lorens, JB
PloS one
4
e5798
2009
Kivonat megmutatása
Blood vessels comprise endothelial cells, mural cells (pericytes/vascular smooth muscle cells) and basement membrane. During angiogenesis, mural cells are recruited to sprouting endothelial cells and define a stabilizing context, comprising cell-cell contacts, secreted growth factors and extracellular matrix components, that drives vessel maturation and resistance to anti-angiogenic therapeutics.To better understand the basis for mural cell regulation of angiogenesis, we conducted high content imaging analysis on a microtiter plate format in vitro organotypic blood vessel system comprising primary human endothelial cells co-cultured with primary human mural cells. We show that endothelial cells co-cultured with mural cells undergo an extensive series of phenotypic changes reflective of several facets of blood vessel formation and maturation: Loss of cell proliferation, pathfinding-like cell migration, branching morphogenesis, basement membrane extracellular matrix protein deposition, lumen formation, anastamosis and development of a stabilized capillary-like network. This phenotypic sequence required endothelial-mural cell-cell contact, mural cell-derived VEGF and endothelial VEGFR2 signaling. Inhibiting formation of adherens junctions or basement membrane structures abrogated network formation. Notably, inhibition of mural cell VEGF expression could not be rescued by exogenous VEGF.These results suggest a unique role for mural cell-associated VEGF in driving vessel formation and maturation. Teljes cikk | 19495422
 |
Contribution of hepatic stellate cells and matrix metalloproteinase 9 in acute liver failure.
Chunli Yan, Ling Zhou, Yuan-Ping Han, Chunli Yan, Ling Zhou, Yuan-Ping Han
Liver international : official journal of the International Association for the Study of the Liver
28
959-71
2008
Kivonat megmutatása
BACKGROUND/AIMS: Fulminant hepatitis or acute liver failure (ALF), initiated by viral infection or hepatic toxin, is a devastating medical complication without effective therapeutic treatment. In this study, we addressed the potential roles of hepatic stellate cells (HSCs) and their produced matrix metalloproteinases (MMPs) in development of ALF. METHODS: Mice were given lipopolysaccharide (LPS) and beta-galactosamine (GA) or carbon tetrachloride to create ALF and establish the association of IL-1, MMP-9, and caspase-3 in acute liver failure. RESULTS: In response to the hepatic toxin, IL-1 and MMP-9 were promptly induced within 1 hour, followed by caspase-3 activation at 2 hours, and dehiscence of sinusoids at 4 hours, and consequent lethality. In contrast, MMP-9 knockout mice were resistant to lethality and absent of caspase-3 activation, demonstrating an MMP-9-dependent activation of caspase in vivo. Further, IL-1-receptor knockout mice were resistant to lethality in MMP-9 dependent manner, indicating a causative relationship. Although many hepatic cells are capable to produce MMP-9 in vitro, HSCs were demonstrated here as the major hepatic cells to express MMP-9 in liver injury. To recapitulate the sinusoidal microenvironment we cultured primary HSCs in 3-dimensional ECM. In response to IL-1, massive MMP-9 was produced by the 3D culture concomitantly with degradation of type-IV collagen. CONCLUSIONS: Based on these evidences, we propose a novel model to highlight the initiation of acute liver failure: IL-1-induced MMPs by HSCs within the space of Disse and thereafter ECM degradation may provoke the collapse of sinusoids, leading parenchymal cell death and loss of liver functions. | 18507761
 |
The effect of functionalized self-assembling peptide scaffolds on human aortic endothelial cell function.
Elsa Genové, Colette Shen, Shuguang Zhang, Carlos E Semino
Biomaterials
26
3341-51
2004
Kivonat megmutatása
A class of designed self-assembling peptide nanofiber scaffolds with more than 99% water content has been shown to be a good biological material for cell culture. Here, we report the functionalization of one of these peptide scaffolds, RAD16-I (AcN-RADARADARADARADA-CONH2), by direct solid phase synthesis extension at the amino terminal with three short-sequence motifs. These motifs are present in two major protein components of the basement membrane, laminin 1 (YIGSR, RYVVLPR) and collagen IV (TAGSCLRKFSTM). These motifs have been previously shown to promote specific biological activities including endothelial cell adhesion, spreading, and tubular formation. Therefore, the generic functionalized peptide developed was AcN-X-GG-RADARADARADARADA-CONH2 with each motif represented by X. We show in this work that these tailor-made peptide scaffolds enhance the formation of confluent cell monolayers of human aortic endothelial cells (HAEC) in culture. Moreover, additional assays designed to evaluate endothelial cell function showed that HAEC monolayers obtained on these scaffolds not only maintained LDL uptake activity but also enhanced nitric oxide release and elevated laminin 1 and collagen IV deposition. These results suggest that this new scaffold provide a better physiological substrate for endothelial cell culture and suggest its further application for biomedical research, cancer biology and regenerative biology. | 15603830
 |