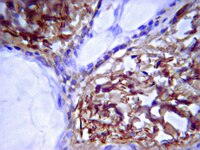
Lateral growth limitation of corneal fibrils and their lamellar stacking depend on covalent collagen cross-linking by transglutaminase-2 and lysyl oxidases, respectively.
Wang, L; Uhlig, PC; Eikenberry, EF; Robenek, H; Bruckner, P; Hansen, U
The Journal of biological chemistry
289
921-9
2014
Kivonat megmutatása
Corneal stroma contains an extracellular matrix of orthogonal lamellae formed by parallel and equidistant fibrils with a homogeneous diameter of ~35 nm. This is indispensable for corneal transparency and mechanical functions. However, the mechanisms controlling corneal fibrillogenesis are incompletely understood and the conditions required for lamellar stacking are essentially unknown. Under appropriate conditions, chick embryo corneal fibroblasts can produce an extracellular matrix in vitro resembling primary corneal stroma during embryonic development. Among other requirements, cross-links between fibrillar collagens, introduced by tissue transglutaminase-2, are necessary for the self-assembly of uniform, small diameter fibrils but not their lamellar stacking. By contrast, the subsequent lamellar organization into plywood-like stacks depends on lysyl aldehyde-derived cross-links introduced by lysyl oxidase activity, which, in turn, only weakly influences fibril diameters. These cross-links are introduced at early stages of fibrillogenesis. The enzymes are likely to be important for a correct matrix deposition also during repair of the cornea. | 24265319
 |
Novel mineral contrast agent for magnetic resonance studies of bone implants grown on a chick chorioallantoic membrane.
Ingrid E Chesnick,Carol B Fowler,Jeffrey T Mason,Kimberlee Potter
Magnetic resonance imaging
29
2010
Kivonat megmutatása
Magnetic resonance imaging (MRI) studies of tissue engineered constructs prior to implantation clearly demonstrate the utility of the MRI technique for studying the bone formation process. To test the utility of our MRI protocols for explant studies, we present a novel test platform in which osteoblast-seeded scaffolds were implanted on the chorioallantoic membrane of a chick embryo. Scaffolds from the following experimental groups were examined by high-resolution MRI: (a) cell-seeded implanted scaffolds (CIM), (b) unseeded implanted scaffolds (UCIM), (c) cell-seeded scaffolds in static culture (CIV) and (d) unseeded scaffolds in static culture (UCIV). The reduction in water proton transverse relaxation times and the concomitant increase in water proton magnetization transfer ratios for CIM and CIV scaffolds, compared to UCIV scaffolds, were consistent with the formation of a bone-like tissue within the polymer scaffold, which was confirmed by immunohistochemistry and fluorescence microscopy. However, the presence of angiogenic vessels and fibrotic adhesions around UCIM scaffolds can confound MRI findings of bone deposition. Consequently, to improve the specificity of the MRI technique for detecting mineralized deposits within explanted tissue engineered bone constructs, we introduce a novel contrast agent that uses alendronate to target a Food and Drug Administration-approved MRI contrast agent (Gd-DOTA) to bone mineral. Our contrast agent termed GdALN was used to uniquely identify mineralized deposits in representative samples from our four experimental groups. After GdALN treatment, both CIM and CIV scaffolds, containing mineralized deposits, showed marked signal enhancement on longitudinal relaxation time-weighted (T1W) images compared to UCIV scaffolds. Relative to UCIV scaffolds, some enhancement was observed in T1W images of GdALN-treated UCIM scaffolds, subjacent to the dark adhesions at the scaffold surface, possibly from dystrophic mineral formed in the fibrotic adhesions. Notably, residual dark areas on T1W images of CIM and UCIM scaffolds were attributable to blood inside infiltrating vessels. In summary, we present the efficacy of GdALN for sensitizing the MRI technique to the deposition of mineralized deposits in explanted polymeric scaffolds. | 21920685
 |
An experimental study on the effect of safflower yellow on tendon injury-repair in chickens.
Liu B, Luo C, Ouyang L, Mu S, Zhu Y, Li K, Zhan M, Liu Z, Jia Y, Lei W
The Journal of surgical research
169
e175-84. Epub 2011 Apr 29.
2010
| 21601885
 |
Macromolecular specificity of collagen fibrillogenesis: fibrils of collagens I and XI contain a heterotypic alloyed core and a collagen I sheath.
Hansen, U; Bruckner, P
The Journal of biological chemistry
278
37352-9
2003
Kivonat megmutatása
Suprastructures of the extracellular matrix, such as banded collagen fibrils, microfibrils, filaments, or networks, are composites comprising more than one type of macromolecule. The suprastructural diversity reflects tissue-specific requirements and is achieved by formation of macromolecular composites that often share their main molecular components alloyed with minor components. Both, the mechanisms of formation and the final macromolecular organizations depend on the identity of the components and their quantitative contribution. Collagen I is the predominant matrix constituent in many tissues and aggregates with other collagens and/or fibril-associated macromolecules into distinct types of banded fibrils. Here, we studied co-assembly of collagens I and XI, which co-exist in fibrils of several normal and pathologically altered tissues, including fibrous cartilage and bone, or osteoarthritic joints. Immediately upon initiation of fibrillogenesis, the proteins co-assembled into alloy-like stubby aggregates that represented efficient nucleation sites for the formation of composite fibrils. Propagation of fibrillogenesis occurred by exclusive accretion of collagen I to yield composite fibrils of highly variable diameters. Therefore, collagen I/XI fibrils strikingly differed from the homogeneous fibrillar alloy generated by collagens II and XI, although the constituent polypeptides of collagens I and II are highly homologous. Thus, the mode of aggregation of collagens into vastly diverse fibrillar composites is finely tuned by subtle differences in molecular structures through formation of macromolecular alloys. | 12869566
 |