401003 Sigma-Aldrich(±)-Ibuprofen - CAS 15687-27-1 - Calbiochem
A nonsteroidal anti-inflammatory drug (NSAID) that acts as a reversible and competitive inhibitor of cyclooxygenase 1 (COX-1) (IC₅₀ = 4.85 µM).
More>> A nonsteroidal anti-inflammatory drug (NSAID) that acts as a reversible and competitive inhibitor of cyclooxygenase 1 (COX-1) (IC₅₀ = 4.85 µM). Less<<(±)-Ibuprofen - CAS 15687-27-1 - Calbiochem MSDS (material safety data sheet) or SDS, CoA and CoQ, dossiers, brochures and other available documents.
同义词: [(±)-2-(4-Isobutylphenyl)-propionic Acid
Recommended Products
概述
| Replacement Information |
|---|
重要规格表
| CAS # | Empirical Formula |
|---|---|
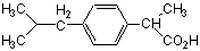
| 15687-27-1 | C₁₃H₁₈O₂ |
价格及供货情况
| 产品目录编号 | 库存情况 | 包装 | 数量 / 包装 | 价格 | 数量 | |
|---|---|---|---|---|---|---|
| 401003-1GMCN |
|
塑胶安瓿;塑胶针药瓶 | 1 gm |
|
— |
| Product Information | |
|---|---|
| CAS number | 15687-27-1 |
| ATP Competitive | Y |
| Form | White solid |
| Hill Formula | C₁₃H₁₈O₂ |
| Chemical formula | C₁₃H₁₈O₂ |
| Reversible | Y |
| Structure formula Image | |
| Quality Level | MQ100 |
| Applications |
|---|
| Biological Information | |
|---|---|
| Primary Target | COX-1 |
| Primary Target IC<sub>50</sub> | 4.85 µM against COX-1 |
| Purity | ≥98% by titration |
| Physicochemical Information | |
|---|---|
| Cell permeable | N |
| Dimensions |
|---|
| Materials Information |
|---|
| Toxicological Information |
|---|
| Safety Information according to GHS | |
|---|---|
| RTECS | MU6640000 |
| Safety Information | |
|---|---|
| R Phrase | R: 22 Harmful if swallowed. |
| S Phrase | S: 36 Wear suitable protective clothing. |
| Product Usage Statements |
|---|
| Packaging Information |
|---|
| Transport Information |
|---|
| Supplemental Information |
|---|
| Specifications |
|---|
| Global Trade Item Number | |
|---|---|
| 产品目录编号 | GTIN |
| 401003-1GMCN | 04055977189377 |
Documentation
(±)-Ibuprofen - CAS 15687-27-1 - Calbiochem MSDS
| 职位 |
|---|
(±)-Ibuprofen - CAS 15687-27-1 - Calbiochem 分析证书
| 标题 | 批号 |
|---|---|
| 401003 |
参考
| 参考信息概述 |
|---|
| Asanuma, M., et al. 2001. J. Neurochem. 76, 1895. Blasko, I., et al. 2001. Neurobiol. Dis. 8, 1094. Ouellet, M., et al. 2001. Proc. Natl. Acad. Sci. USA 98, 14583. Casper, D., et al. 2000. Neurosci. Lett. 289, 201. Lambat, Z., et al. 2000. Metab. Brain Dis. 15, 249. Lim, G.P., et al. 2000. J. Neurosci. 20, 5709. Ogawa, O., et al. 2000. Eur. J. Pharmacol. 408, 137. Wyss-Coray, T., and Mucke, L. 2000. Nat. Med. 6, 973. Lehmann, J.M., et al. 1997. J. Biol. Chem. 272, 3406. Boneburg, E.M., et al. 1996. J. Clin. Pharmacol. 36, 16S. Mitchell, J.A., et al. 1994. Proc. Natl. Acad. Sci. USA 90, 11693. |