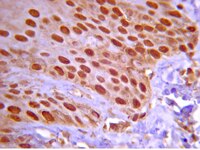
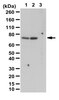
Midkine promotes neuroblastoma through Notch2 signaling.
Kishida, S; Mu, P; Miyakawa, S; Fujiwara, M; Abe, T; Sakamoto, K; Onishi, A; Nakamura, Y; Kadomatsu, K
Cancer research
73
1318-27
2013
Show Abstract
Midkine is a heparin-binding growth factor highly expressed in various cancers, including neuroblastoma, the most common extracranial pediatric solid tumor. Prognosis of patients with neuroblastoma in which MYCN is amplified remains particularly poor. In this study, we used a MYCN transgenic model for neuroblastoma in which midkine is highly expressed in precancerous lesions of sympathetic ganglia. Genetic ablation of midkine in this model delayed tumor formation and reduced tumor incidence. Furthermore, an RNA aptamer that specifically bound midkine suppressed the growth of neuroblastoma cells in vitro and in vivo in tumor xenografts. In precancerous lesions, midkine-deficient MYCN transgenic mice exhibited defects in activation of Notch2, a candidate midkine receptor, and expression of the Notch target gene HES1. Similarly, RNA aptamer-treated tumor xenografts also showed attenuation of Notch2-HES1 signaling. Our findings establish a critical role for the midkine-Notch2 signaling axis in neuroblastoma tumorigenesis, which implicates new strategies to treat neuroblastoma. | Immunohistochemistry | 23243020
 |
Notch signaling components are upregulated during both endochondral and intramembranous bone regeneration.
Dishowitz, MI; Terkhorn, SP; Bostic, SA; Hankenson, KD
Journal of orthopaedic research : official publication of the Orthopaedic Research Society
30
296-303
2012
Show Abstract
Previous studies have demonstrated that Notch signaling regulates endochondral and intramembranous bone formation by controlling cell proliferation and differentiation. Notch signaling has also been shown to regulate healing in a variety of tissues. The objective of this study was to characterize and compare activation of the Notch signaling pathway during endochondral and intramembranous bone healing using tibial fracture and calvarial defect injury models, respectively. Bilateral tibial fractures or bilateral 1.5 mm diameter calvarial defects were created in mice, and tissues were harvested at 0, 5, 10, and 20 days post-fracture. Gene expression of Notch signaling components was upregulated during both tibial fracture and calvarial defect healing, with expression generally higher during tibial fracture healing. The most highly expressed ligand and receptor during healing, Jag1 and Notch2 (specifically the activated receptor, known as NICD2), were similarly localized in mesenchymal cells during both modes of healing, with expression decreasing during chondrogenesis, but remaining present in osteoblasts at all stages of maturity. Results suggest that in addition to embryological bone development, Notch signaling regulates both endochondral and intramembranous bone healing. | Immunohistochemistry (Paraffin) | 21818769
 |