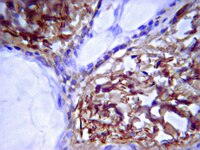
Control of somatic tissue differentiation by the long non-coding RNA TINCR.
Kretz, M; Siprashvili, Z; Chu, C; Webster, DE; Zehnder, A; Qu, K; Lee, CS; Flockhart, RJ; Groff, AF; Chow, J; Johnston, D; Kim, GE; Spitale, RC; Flynn, RA; Zheng, GX; Aiyer, S; Raj, A; Rinn, JL; Chang, HY; Khavari, PA
Nature
493
231-5
2013
Afficher le résumé
Several of the thousands of human long non-coding RNAs (lncRNAs) have been functionally characterized; however, potential roles for lncRNAs in somatic tissue differentiation remain poorly understood. Here we show that a 3.7-kilobase lncRNA, terminal differentiation-induced ncRNA (TINCR), controls human epidermal differentiation by a post-transcriptional mechanism. TINCR is required for high messenger RNA abundance of key differentiation genes, many of which are mutated in human skin diseases, including FLG, LOR, ALOXE3, ALOX12B, ABCA12, CASP14 and ELOVL3. TINCR-deficient epidermis lacked terminal differentiation ultrastructure, including keratohyalin granules and intact lamellar bodies. Genome-scale RNA interactome analysis revealed that TINCR interacts with a range of differentiation mRNAs. TINCR-mRNA interaction occurs through a 25-nucleotide 'TINCR box' motif that is strongly enriched in interacting mRNAs and required for TINCR binding. A high-throughput screen to analyse TINCR binding capacity to approximately 9,400 human recombinant proteins revealed direct binding of TINCR RNA to the staufen1 (STAU1) protein. STAU1-deficient tissue recapitulated the impaired differentiation seen with TINCR depletion. Loss of UPF1 and UPF2, both of which are required for STAU1-mediated RNA decay, however, did not have differentiation effects. Instead, the TINCR-STAU1 complex seems to mediate stabilization of differentiation mRNAs, such as KRT80. These data identify TINCR as a key lncRNA required for somatic tissue differentiation, which occurs through lncRNA binding to differentiation mRNAs to ensure their expression. | | 23201690
 |
Laminin-511 and integrin beta-1 in hair follicle development and basal cell carcinoma formation.
DeRouen, MC; Zhen, H; Tan, SH; Williams, S; Marinkovich, MP; Oro, AE
BMC developmental biology
10
112
2009
Afficher le résumé
Initiation of the hair follicle placode and its subsequent growth, maturation and cycling in post-natal skin requires signaling interactions between epithelial cells and adjacent dermal cells and involves Shh signaling via the primary cilium. Previous reports have implicated laminins in hair follicle epithelial invagination.Here we use a human BCC model system and mouse mutants to re-evaluate the role of laminin-511 in epithelial invagination in the skin. Blocking laminin 511 and 332 in BCCs maintains primary cilia and Shh signalling, but prevents invagination. Similarly, in laminin-511 and dermal beta-1 integrin mutants, dermal papilla development and primary cilia formation are normal. Dermal beta-1 integrin mutants have normal hair follicle development.Our data provides support for a primary role of laminin-511 promoting hair follicle epithelial downgrowth without affecting dermal primary cilia and Shh target gene induction. Article en texte intégral | | 21067603
 |
The development and characterization of an in vitro model of psoriasis.
Barker, CL; McHale, MT; Gillies, AK; Waller, J; Pearce, DM; Osborne, J; Hutchinson, PE; Smith, GM; Pringle, JH
The Journal of investigative dermatology
123
892-901
2004
Afficher le résumé
In this study, the phenotype of psoriatic keratinocytes and fibroblasts in reconstructed skin models was compared to those constructed from normal cells. Characterization of this model by immunohistochemistry showed that classical markers of keratinocyte differentiation exhibited similar patterns of distribution in the psoriatic models to those derived from normal cells and generally reflected in vivo observations. Some crucial differences, however, were observed between normal and psoriatic models when pro-inflammatory gene expression and keratinocyte proliferation were investigated. Notably, the chemokine receptor CXCR2 was overexpressed in the psoriatic models, and, moreover, was localized to the granular layer of keratinocytes as seen in psoriasis in vivo. Pro-inflammatory genes (tumor necrosis factor alpha [TNF-alpha], interferon gamma [IFN-gamma], and interleukin 8 [IL-8]) were expressed at high levels in the psoriatic models, but were only minimally expressed in the normal models. Models derived from uninvolved psoriatic skin showed the same gene expression profile as those derived from involved skin along with an increased proliferation rate when compared to normal models. These results suggest that psoriatic individuals possess an inherent predisposition to develop the disease phenotype even in the absence of T cells. This study represents a comprehensive characterization of psoriatic human skin reconstructed in vitro, and demonstrates the potential of this model as a valuable tool in drug discovery. | Immunohistochemistry | 15482477
 |