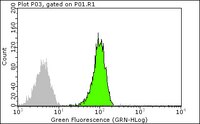
17-218 Sigma-AldrichRas Activation Assay Kit

More>>
Less<<
Key Applications:
Enzyme Assays
Ras Activation Assay Kit: SDB (Sicherheitsdatenblätter), Analysenzertifikate und Qualitätszertifikate, Dossiers, Broschüren und andere verfügbare Dokumente.
Übersicht
Key Spec Table
| Key Applications |
|---|
| Enzyme Assays |
| Description | |
|---|---|
| Catalogue Number | 17-218 |
| Replaces | SGT435 |
| Brand Family | Upstate |
| Trade Name |
|
| Description | Ras Activation Assay Kit |
| Product Information | |
|---|---|
| Components |
|
| Quality Level | MQ100 |
| Applications | |
|---|---|
| Key Applications |
|
| Packaging Information | |
|---|---|
| Material Size | 1 kit |
| Material Package | Kit capacity: 30 assays |
| Global Trade Item Number | |
|---|---|
| Bestellnummer | GTIN |
| 17-218 | 04053252516689 |
Documentation
Ras Activation Assay Kit SDB
| Titel |
|---|
Ras Activation Assay Kit Analysenzertifikate
Literatur
| Übersicht | Anwendung | Pub Med ID |
|---|---|---|
| The TGFβ receptor-interacting protein km23-1/DYNLRB1 plays an adaptor role in TGFβ1 autoinduction via its association with Ras. Jin, Q; Ding, W; Mulder, KM The Journal of biological chemistry 287 26453-63 2011 Abstract anzeigen We have previously elucidated the signaling events that are required for TGFβ1 autoinduction (Yue, J., and Mulder, K. M. (2000) J. Biol. Chem. 275, 30765-30773). Further, we have reported that the TGFβ receptor (TβR)-interacting protein km23-1 plays an important role in TGFβ signal transduction (Jin, Q., Ding, W., and Mulder, K. M. (2007) J. Biol. Chem. 282, 19122-19132). Here we examined the role of km23-1 in TGFβ1 autoinduction in TGFβ-sensitive epithelial cells. siRNA blockade of km23-1 reduced TGFβ1 mRNA expression, as well as DNA binding and transcriptional activation of the relevant activator protein-1 site in the human TGFβ1 promoter. Further, knockdown of km23-1 inhibited TGFβ-mediated activation of ERK and JNK, phosphorylation of c-Jun, and transactivation of the c-Jun promoter. Sucrose gradient analyses indicate that km23-1 was present in lipid rafts together with Ras and TβRII after TGFβ treatment. Immunoprecipitation/blot analyses revealed the formation of a TGFβ-inducible complex between Ras and km23-1 in vivo within minutes of TGFβ addition. Moreover, we demonstrate for the first time that km23-1 is required for Ras activation by TGFβ. Our results indicate that km23-1 is required for TGFβ1 autoinduction through Smad2-independent Ras/ERK/JNK pathways. More importantly, our findings demonstrate that km23-1 functions as a critical adaptor coupling TβR activation to activation of Ras effector pathways downstream. | 22637579
 | |
| E4orf1: a novel ligand that improves glucose disposal in cell culture. Dhurandhar, EJ; Dubuisson, O; Mashtalir, N; Krishnapuram, R; Hegde, V; Dhurandhar, NV PloS one 6 e23394 2010 Abstract anzeigen Reducing dietary fat intake and excess adiposity, the cornerstones of behavioral treatment of insulin resistance (IR), are marginally successful over the long term. Ad36, a human adenovirus, offers a template to improve IR, independent of dietary fat intake or adiposity. Ad36 increases cellular glucose uptake via a Ras-mediated activation of phosphatidyl inositol 3-kinase(PI3K), and improves hyperglycemia in mice, despite a high-fat diet and without reducing adiposity. Ex-vivo studies suggest that Ad36 improves hyperglycemia in mice by increasing glucose uptake by adipose tissue and skeletal muscle, and by reducing hepatic glucose output. It is impractical to use Ad36 for therapeutic action. Instead, we investigated if the E4orf1 protein of Ad36, mediates its anti-hyperglycemic action. Such a candidate protein may offer an attractive template for therapeutic development. Experiment-1 determined that Ad36 'requires' E4orf1 protein to up-regulate cellular glucose uptake. Ad36 significantly increased glucose uptake in 3T3-L1 preadipocytes, which was abrogated by knocking down E4orf1 with siRNA. Experiment-2 identified E4orf1 as 'sufficient' to up-regulate glucose uptake. 3T3-L1 cells that inducibly express E4orf1, increased glucose uptake in an induction-dependent manner, compared to null vector control cells. E4orf1 up-regulated PI3K pathway and increased abundance of Ras--the obligatory molecule in Ad36-induced glucose uptake. Experiment-3: Signaling studies of cells transiently transfected with E4orf1 or a null vector, revealed that E4orf1 may activate Ras/PI3K pathway by binding to Drosophila discs-large (Dlg1) protein. E4orf1 activated total Ras and, particularly the H-Ras isoform. By mutating the PDZ domain binding motif (PBM) of E4orf1, Experiment-4 showed that E4orf1 requires its PBM to increase Ras activation or glucose uptake. Experiment-5: In-vitro, a transient transfection by E4orf1 significantly increased glucose uptake in preadipocytes, adipocytes, or myoblasts, and reduced glucose output by hepatocytes. Thus, the highly attractive anti-hyperglycemic effect of Ad36 is mirrored by E4orf1 protein, which may offer a novel ligand to develop anti-hyperglycemic drugs. Volltextartikel | 21886789
 | |
| Effects of estrogen on stress-induced premature senescence of vascular smooth muscle cells: A novel mechanism for the \time window theory\ of menopausal hormone therapy. Zhu C, Zhang L, Zheng Y, Xu J, Song J, Rolfe BE, Campbell JH Atherosclerosis 215 294-300. Epub 2011 Jan 19. 2010 | 21300357
 | |
| Ligand-specific function of transforming growth factor beta in epithelial-mesenchymal transition in heart development. Azhar, M; Runyan, RB; Gard, C; Sanford, LP; Miller, ML; Andringa, A; Pawlowski, S; Rajan, S; Doetschman, T Developmental dynamics : an official publication of the American Association of Anatomists 238 431-42 2009 Abstract anzeigen The ligand specificity of transforming growth factor beta (TGFbeta) in vivo in mouse cardiac cushion epithelial-to-mesenchymal transition (EMT) is poorly understood. To elucidate the function of TGFbeta in cushion EMT, we analyzed Tgfb1(-/-), Tgfb2(-/-), and Tgfb3(-/-) mice between embryonic day (E) 9.5 and E14.5 using both in vitro and in vivo approaches. Atrioventricular (AV) canal collagen gel assays at E9.5 indicated normal EMT in both Tgfb1(-/-) and Tgfb3(-/-) mice. However, analysis of Tgfb2(-/-) AV explants at E9.5 and E10.5 indicated that EMT, but not cushion cell proliferation, was initially delayed but later remained persistent. This was concordant with the observation that Tgfb2(-/-) embryos, and not Tgfb1(-/-) or Tgfb3(-/-) embryos, develop enlarged cushions at E14.5 with elevated levels of well-validated indicators of EMT. Collectively, these data indicate that TGFbeta2, and not TGFbeta1 or TGFbeta3, mediates cardiac cushion EMT by promoting both the initiation and cessation of EMT. Volltextartikel | 19161227
 | |
| Gene silencing for epidermal growth factor receptor variant III induces cell-specific cytotoxicity. Yamoutpour, Farnaz, et al. Mol. Cancer Ther., 7: 3586-97 (2008) 2008 Abstract anzeigen Epidermal growth factor receptor variant III (EGFRvIII) is a constitutively active mutant form of EGFR that is expressed in 40% to 50% of gliomas and several other malignancies. Here, we describe the therapeutic effects of silencing EGFRvIII on glioma cell lines in vitro and in vivo. A small interfering RNA molecule against EGFRvIII was introduced into EGFRvIII-expressing glioma cells (U87Delta) by electroporation resulting in complete inhibition of expression of EGFRvIII as early as 48 h post-treatment. During EGFRvIII silencing, a decrease in the proliferation and invasiveness of U87Delta cells was accompanied by an increase in apoptosis (P < 0.05). Notably, EGFRvIII silencing inhibited the signal transduction machinery downstream of EGFRvIII as evidenced by decreases in the activated levels of Ras and extracellular signal-regulated kinase. A lentivirus capable of expressing anti-EGFRvIII short hairpin RNA was also able to achieve progressive silencing of EGFRvIII in U87Delta cells in addition to inhibiting cell proliferation, invasiveness, and colony formation in a significant manner (P < 0.05). Silencing EGFRvIII in U87Delta cultures with this virus reduced the expression of factors involved in epithelial-mesenchymal transition including N-cadherin, beta-catenin, Snail, Slug, and paxillin but not E-cadherin. The anti-EGFRvIII lentivirus also affected the cell cycle progression of U87Delta cells with a decrease in G(1) and increase in S and G(2) fractions. In an in vivo model, tumor growth was completely inhibited in severe combined immunodeficient mice (n = 10) injected s.c. with U87Delta cells treated with the anti-EGFRvIII lentivirus (P = 0.005). We conclude that gene specific silencing of EGFRvIII is a promising strategy for treating cancers that contain this mutated receptor. | 19001441
 | |
| Activated K-RAS increases polyamine uptake in human colon cancer cells through modulation of caveolar endocytosis. Upal K Basu Roy,Nathaniel S Rial,Karen L Kachel,Eugene W Gerner Molecular carcinogenesis 47 2008 Abstract anzeigen Endocytic pathways have been implicated in polyamine transport in mammalian cells, but specific mechanisms have not been described. We have shown that expression of a dominant negative (DN) form of the GTPase Dynamin, but not Eps15, diminished polyamine uptake in colon cancer cells indicating a caveolar and nonclathrin uptake mode. Polyamines co-sediment with lipid raft/caveolin-1 rich fractions, of the plasma membrane in a sucrose density gradient. Knock down of caveolin-1 significantly increased polyamine uptake. Conversely, ectopic expression of this protein resulted in diminished polyamine uptake. We also found that presence of an activated K-RAS oncogene significantly increased polyamine uptake by colon cancer cells. This effect is through an increase in caveolin-1 phosphorylation at tyrosine residue 14. Caveolin-1 is a negative regulator of caveolar endocytosis and phosphorylation in a K-RAS dependent manner leads to an increase in caveolar endocytosis. In cells expressing wild type K-RAS, addition of exogenous uPA was sufficient to stimulate caveolar endocytosis of polyamines. This effect was abrogated by the addition of a SRC kinase inhibitor. These data indicate that polyamine transport follows a dynamin-dependent and clathrin-independent endocytic uptake route, and this route is positively regulated by the oncogenic expression of K-RAS in a caveolin-1 dependent manner. Volltextartikel | 18176934
 | |
| RIG1 suppresses Ras activation and induces cellular apoptosis at the Golgi apparatus. Tsai, Fu-Ming, et al. Cell. Signal., 19: 989-99 (2007) 2007 Abstract anzeigen Retinoid-inducible gene 1 encodes RIG1 is a growth regulator, which inhibits the pathways of the RAS/mitogen-activated protein kinases by suppressing the activation of RAS. Confocal microscopic analysis demonstrated that RIG1 is localized in the endoplasmic reticulum (ER) and Golgi apparatus in HtTA cervical cancer cells. Carboxyterminal-deleted RIG1 targeted to the Golgi or ER was constructed and validated. The activation of HRAS was inhibited by 25.1% or 81.4% in cells cotransfected with wild-type or Golgi-targeted RIG1, respectively. Expression of wild-type or Golgi-targeted RIG1 for 24 h induced cellular apoptosis in HtTA cells, as assessed by MTT assay, the release of lactate dehydrogenase, and chromatin condensation. In contrast, ER-targeted RIG1 and carboxyterminal-deleted RIG1 (RIG1DeltaC) exhibited no activity. Caspase-2, -3, and -9 were activated following the expression of wild-type and Golgi-targeted RIG1. Although the caspase-3 inhibitor Z-DEVD-FMK partially or completely reversed the cell death induced by wild-type or Golgi-targeted RIG1, it did not prevent the anti-RAS effect of RIG1. In conclusion, the proapoptotic and anti-RAS activities of RIG1 are primarily associated with the Golgi localization of the protein. The proapoptotic activities of RIG1 are mediated through the activation of caspase-2 and -3 and are independent of its effect on RAS. | 17196792
 | |
| RIG1 inhibits the Ras/mitogen-activated protein kinase pathway by suppressing the activation of Ras. Tsai, Fu-Ming, et al. Cell. Signal., 18: 349-58 (2006) 2005 Abstract anzeigen The retinoid-inducible gene 1 (RIG1) protein is a retinoid-inducible growth regulator. Previous studies have shown that the RIG1 protein inhibits the signaling pathways of Ras/mitogen-activated protein kinases. However, neither the mode of action nor the site of inhibition of RIG1 is known. This study investigated the effects of RIG1, and the mechanisms responsible for these effects, on the activation of Ras proteins in HtTA cervical cancer cells. RIG1 reduced the levels of activated Ras (Ras-GTP) and total Ras protein in cells transfected with mutated H-, N-, or K-Ras(G12V), or in cells transfected with the wild type H- or N-Ras followed by stimulation with epidermal growth factor. The half-life of Ras protein decreased from more than 36 h in control cells to 18 h in RIG1-transfected cells. RIG1 immunoprecipitated with the Ras protein in co-transfected cellular lysates. In contrast to the predominant plasma membrane localization in control cells, the H-Ras fusion protein EGFP-H-Ras was localized within a discrete cytoplasmic compartment where it co-localized with RIG1. RIG1 inhibited more than 93% of the Elk- and CHOP-mediated transactivation induced by H- or K-Ras(G12V). However, RIG1 did not inhibit the transactivation induced by MEK1 or MEK3, and failed to suppress the phosphorylation of extracellular signal-regulated kinases 1 and 2 induced by the constitutively activated B-Raf(V599E). The RIG1 with carboxyl terminal truncation (RIG1DeltaC) did not immunoprecipitate with Ras and had no effect on Ras activation or transactivation of the downstream signal pathways. These data indicate that RIG1 exerts its inhibitory effect at the level of Ras activation, which is independent of Ras subtype but dependent on the membrane localization of the RIG1 protein. This inhibition of Ras activation may be mediated through downregulation of Ras levels and alteration of Ras subcellular distribution. | 16005186
 | |
| Optimization of a nonradioactive method for consistent and sensitive determination of activated K-ras protein. Richard J Calvert,Wafa Kammouni,Keith D Kikawa Analytical biochemistry 343 2004 Abstract anzeigen Accurate measurement of activity of wild-type K-ras protein is important due to its tumor suppressor action in tissues such as lung. A published method by Taylor and co-workers uses plasmid-containing Escherichia coli cells to produce a glutathione-S-transferase/raf-1 ras binding domain (GST-RBD) fusion protein attached to glutathione beads to isolate activated ras protein. We systematically optimized the method before use on lung tissues. Changing the GST-RBD protein induction temperature from the original 37 to 30 degrees C produced a consistently greater yield of fusion protein. To improve stability of the GST-RBD beads so as to perform large-scale experiments, 0.1% NaN(3) was added. NaN(3)-treated beads retained full affinity for at least 24 days. Sensitivity was improved by using a polyvinylidene difluoride membrane rather than nitrocellulose for immunoblotting. We also compared our GST-RBD beads with two commercial assay kits and found that our beads had both superior sensitivity and reduced variability. In summary, our modification of the GST-RBD affinity method to recover activated K-ras greatly increased the yield of fusion protein, prolonged the useful life of GST-RBD beads to at least 24 days, and enhanced detection sensitivity. | 16018961
 | |
| Methylation and inhibition of expression of uPA by the RAS oncogene: divergence of growth control and invasion in breast cancer cells. Pouya Pakneshan, Moshe Szyf, Shafaat A Rabbani Carcinogenesis 26 557-64 2004 Abstract anzeigen Expression of urokinase-type plasminogen activator (uPA), a protease only expressed in highly invasive human breast cancer cells, is inhibited by DNA methylation of its promoter. We tested the hypothesis that up-regulation of DNA methyltransferase 1 (DNMT1) will lead to methylation and silencing of uPA and inhibition of the invasiveness of metastatic breast cancer cells. Since RAS was previously shown to up-regulate DNA methylation, we examined the effects of ectopic expression of constitutively active RAS on methylation and expression of uPA. Transfection of Ha-RAS into MDA-MB-231 human breast cancer cells resulted in a significantly shorter cell doubling time compared with the controls. However, expression and activity of the metastatic gene uPA and invasive capacity of the cells were significantly reduced by the oncogene RAS. Silencing of uPA by RAS is mediated by a cis modification of the uPA promoter and not through an effect on a trans-acting factor, since a transiently transfected unmethylated uPA-luicferase reporter is expressed at a similar level in RAS-transfected and control cells. We then examined the levels of DNMT1 and methylated DNA-binding protein 2 (MBD2) expressions in these cells to determine whether this reduction in uPA expression is associated with changes in the DNA methylation machinery. Our results showed that ectopic expression of RAS induced DNMT1 expression and activity and inhibited MBD2 expression. Consistent with methylation-mediated repression, uPA was partially methylated in RAS-transfected cells and uPA expression was induced upon treatment of RAS transfectants with the demethylating agent 5'-azacytidine. These results therefore imply that the RAS-DNMT1 DNA methylation pathway which promotes oncogenic growth in many cancers can exert an opposite effect on the invasive capacity of the highly invasive MDA-MB-231 cells, thus illustrating the divergence of growth and metastasis promoting pathways in cancer. This has important implications for new therapeutic approaches to metastasizing cancer. | 15618232
 |
Broschüre
| Titel |
|---|
| Hallmarks of Aging |
Verwandte Produkte & Anwendungen
Produktfamilien
RAS Antibodies, Kits, Proteins & PlasmidsMillipore’s RAS antibodies, kits, and plasmids are high quality, validated tools for research. These are based on the expertise of Upstate & Chemicon. See below for Millipore’s Ras G-protein products.Weitere Informationen >> |
Kategorien
| Life Science Research > Protein Detection and Quantification > Activity Assays > Activation Assays |