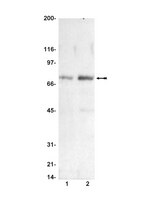
A genome-scale RNA-interference screen identifies RRAS signaling as a pathologic feature of Huntington's disease.
Miller, JP; Yates, BE; Al-Ramahi, I; Berman, AE; Sanhueza, M; Kim, E; de Haro, M; DeGiacomo, F; Torcassi, C; Holcomb, J; Gafni, J; Mooney, SD; Botas, J; Ellerby, LM; Hughes, RE
PLoS genetics
8
e1003042
2012
显示摘要
A genome-scale RNAi screen was performed in a mammalian cell-based assay to identify modifiers of mutant huntingtin toxicity. Ontology analysis of suppressor data identified processes previously implicated in Huntington's disease, including proteolysis, glutamate excitotoxicity, and mitochondrial dysfunction. In addition to established mechanisms, the screen identified multiple components of the RRAS signaling pathway as loss-of-function suppressors of mutant huntingtin toxicity in human and mouse cell models. Loss-of-function in orthologous RRAS pathway members also suppressed motor dysfunction in a Drosophila model of Huntington's disease. Abnormal activation of RRAS and a down-stream effector, RAF1, was observed in cellular models and a mouse model of Huntington's disease. We also observe co-localization of RRAS and mutant huntingtin in cells and in mouse striatum, suggesting that activation of R-Ras may occur through protein interaction. These data indicate that mutant huntingtin exerts a pathogenic effect on this pathway that can be corrected at multiple intervention points including RRAS, FNTA/B, PIN1, and PLK1. Consistent with these results, chemical inhibition of farnesyltransferase can also suppress mutant huntingtin toxicity. These data suggest that pharmacological inhibition of RRAS signaling may confer therapeutic benefit in Huntington's disease. | Western Blotting | | 23209424
 |
RIN1 orchestrates the activation of RAB5 GTPases and ABL tyrosine kinases to determine the fate of EGFR.
Balaji, K; Mooser, C; Janson, CM; Bliss, JM; Hojjat, H; Colicelli, J
Journal of cell science
125
5887-96
2012
显示摘要
Stimulation of epidermal growth factor receptor (EGFR) initiates RAS signaling simultaneously with EGFR internalization. Endocytosed EGFR is then either recycled or degraded. EGFR fate is determined in part by the RAS effector RIN1, a guanine nucleotide exchange factor (GEF) for RAB5 GTPases. EGFR degradation was slowed by RIN1 silencing, enhanced by RIN1 overexpression and accelerated by RIN1 localization to the plasma membrane. RIN1 also directly activates ABL tyrosine kinases, which regulate actin remodeling, a function not previously connected to endocytosis. We report that RIN1-RAB5 signaling favors EGFR downregulation over EGFR recycling, whereas RIN1-ABL signaling stabilizes EGFR and inhibits macropinocytosis. RIN1(QM), a mutant that blocks ABL activation, caused EGF-stimulated membrane ruffling, actin remodeling, dextran uptake and EGFR degradation. An ABL kinase inhibitor phenocopied these effects in cells overexpressing RIN1. EGFR activation also promotes RIN1 interaction with BIN1, a membrane bending protein. These findings suggest that RIN1 orchestrates RAB5 activation, ABL kinase activation and BIN1 recruitment to determine EGFR fate. | Western Blotting | Human | 22976291
 |
Regulation of mTORC1 signaling by pH.
Balgi, AD; Diering, GH; Donohue, E; Lam, KK; Fonseca, BD; Zimmerman, C; Numata, M; Roberge, M
PloS one
6
e21549
2011
显示摘要
Acidification of the cytoplasm and the extracellular environment is associated with many physiological and pathological conditions, such as intense exercise, hypoxia and tumourigenesis. Acidification affects important cellular functions including protein synthesis, growth, and proliferation. Many of these vital functions are controlled by mTORC1, a master regulator protein kinase that is activated by various growth-stimulating signals and inactivated by starvation conditions. Whether mTORC1 can also respond to changes in extracellular or cytoplasmic pH and play a role in limiting anabolic processes in acidic conditions is not known.We examined the effects of acidifying the extracellular medium from pH 7.4 to 6.4 on human breast carcinoma MCF-7 cells and immortalized mouse embryo fibroblasts. Decreasing the extracellular pH caused intracellular acidification and rapid, graded and reversible inhibition of mTORC1, assessed by measuring the phosphorylation of the mTORC1 substrate S6K. Fibroblasts deleted of the tuberous sclerosis complex TSC2 gene, a major negative regulator of mTORC1, were unable to inhibit mTORC1 in acidic extracellular conditions, showing that the TSC1-TSC2 complex is required for this response. Examination of the major upstream pathways converging on the TSC1-TSC2 complex showed that Akt signaling was unaffected by pH but that the Raf/MEK/ERK pathway was inhibited. Inhibition of MEK with drugs caused only modest mTORC1 inhibition, implying that other unidentified pathways also play major roles.This study reveals a novel role for the TSC1/TSC2 complex and mTORC1 in sensing variations in ambient pH. As a common feature of low tissue perfusion, low glucose availability and high energy expenditure, acidic pH may serve as a signal for mTORC1 to downregulate energy-consuming anabolic processes such as protein synthesis as an adaptive response to metabolically stressful conditions. 全文本文章 | | | 21738705
 |
BRAF(V600E) efficient transformation and induction of microsatellite instability versus KRAS(G12V) induction of senescence markers in human colon cancer cells.
Oikonomou, E; Makrodouli, E; Evagelidou, M; Joyce, T; Probert, L; Pintzas, A
Neoplasia (New York, N.Y.)
11
1116-31
2009
显示摘要
In colorectal cancer, BRAF and KRAS oncogenes are mutated in about 15% and 35% respectively at approximately the same stage of the adenoma-carcinoma sequence. Since these two mutations rarely coexist, further analysis to dissect their function of transformation in colon cancer is required. Caco-2 human colon adenocarcinoma cells were stably transfected with BRAF(V600E) (Caco-BR cells) or KRAS(G12V) (Caco-K cells) oncogenes. BRAF(V600E) is more efficient in transforming Caco-2 cells and altering their morphology. The dominant nature of BRAF(V600E) is evident by its ability to render Caco-2 cells tumorigenic in vivo all be it through selective extracellular signal-related kinase (ERK) 2 phosphorylation and high levels of cyclin D1. As a consequence, the cell cycle distribution of parental cells is altered and microsatellite instability is introduced. Attenuated ERK activation observed correlated with KSR downregulation by BRAF(V600E) without further implications to signaling. Highly activated ERK in case of KRAS(G12V) (Caco-K cells) leads to mild transformation causing Caco-K cells to express premature senescence-related markers and acquire growth factor-dependent viability. Interestingly, BRAF(WT)gets equally activated by upstream KRAS mutations present in colon adenocarcinoma cells such as DLD-1 and SW620. Taken together, these results suggest that the two oncogenes have different transforming capability in colon cancer, although they both use the mitogen-activated protein (MAP) kinase pathway to carry out their effect. In general, BRAF(V600E) presents greater potential in mediating tumorigenic effect as compared to KRAS(G12V) both in vivo and in vitro. These findings may have implications in personalised diagnosis and targeted therapeutics. | Western Blotting | | 19881948
 |
Filamin A-mediated down-regulation of the exchange factor Ras-GRF1 correlates with decreased matrix metalloproteinase-9 expression in human melanoma cells.
Zhu, TN; He, HJ; Kole, S; D'Souza, T; Agarwal, R; Morin, PJ; Bernier, M
The Journal of biological chemistry
282
14816-26
2007
显示摘要
The actin-binding protein filamin A (FLNa) is associated with diverse cellular processes such as cell motility and signaling through its scaffolding properties. Here we examine the effect of FLNa on the regulation of signaling pathways that control the expression of matrix metalloproteinases (MMPs). The lack of FLNa in human M2 melanoma cells was associated with constitutive and phorbol ester-induced expression and secretion of active MMP-9 in the absence of MMP-2 up-regulation. M2 cells displayed stronger MMP-9 production and activity than their M2A7 counterparts where FLNa had been stably reintroduced. Using an MMP-9 promoter construct (pMMP-9-Luc), in vitro kinase assays, and genetic and pharmacological approaches, we demonstrate that FLNa mediated transcriptional down-regulation of pMMP-9-Luc by suppressing the constitutive hyperactivity of the Ras/MAPK extracellular signal-regulated kinase (ERK) cascade. Experimental evidence indicated that this phenomenon was associated with destabilization and ubiquitylation of Ras-GRF1, a guanine nucleotide exchange factor that activates H-Ras by facilitating the release of GDP. Ectopic expression of Ras-GRF1 was accompanied by ERK activation and elevated levels of MMP-9 in M2A7 cells, whereas a catalytically inactive dominant negative Ras-GRF1, which prevented ERK activation, reduced MMP-9 expression in M2 cells. Our results indicate that expression of FLNa regulates constitutive activation of the Ras/ERK pathway partly through a Ras-GRF1 mechanism to modulate the production of MMP-9. | | | 17389601
 |
Development of a highly reproducible three-dimensional organotypic model of the oral mucosa.
Anna Dongari-Bagtzoglou, Helena Kashleva
Nature protocols
1
2012-8
2006
显示摘要
In this report we describe the development of a standardized three-dimensional (3D) system of the human oral mucosa based on an immortalized human oral keratinocyte cell line (OKF6/TERT-2). The procedure takes approximately 2-3 weeks to complete and includes three main stages: preparation of collagen-embedded fibroblasts, addition of the mucosal component and airlifting of cultures to ensure adequate differentiation/stratification. This procedure results in a multilayer epithelial structure in which layers are organized similarly to the cells in native oral mucosa. Specifically, this model system consists of a stratum basale, having one layer of columnar to round cells, a relatively flattened stratum spinosum and stratum granulosum, and a non-keratinizing stratum corneum. This 3D system resembles the commercially available system based on the cell line TR146 (SkinEthic), with the exception that our model system does not contain dyskeratotic changes and has a submucosal component, and thus better represents the normal human mucosa and submucosa. 全文本文章 | | | 17487190
 |
Raf-1 serine 338 phosphorylation plays a key role in adhesion-dependent activation of extracellular signal-regulated kinase by epidermal growth factor.
Edin, ML; Juliano, RL
Molecular and cellular biology
25
4466-75
2005
显示摘要
Activation of the extracellular signal-regulated kinase (ERK) 1/2 cascade by polypeptide growth factors is tightly coupled to adhesion to extracellular matrix in nontransformed cells. Raf-1, the initial kinase in this cascade, is intricately regulated by phosphorylation, localization, and molecular interactions. We investigated the complex interactions between Raf-1, protein kinase A (PKA), and p21-activated kinase (PAK) to determine their roles in the adhesion dependence of signaling from epidermal growth factor (EGF) to ERK. We conclude that Raf-1 phosphorylation on serine 338 (S338) is a critical step that is inhibited in suspended cells. Restoration of phosphorylation at S338, either by expression of highly active PAK or by expression of an S338 phospho-mimetic Raf-1 mutation, led to a partial rescue of ERK activation in suspended cells. Raf-1 inhibition in suspension was not due to excessive negative regulation on inhibitory sites S43 and S259, as these serines were largely dephosphorylated in suspended cells. Finally, strong phosphorylation of Raf-1 S338 provided resistance to PKA-mediated inhibition of ERK activation. Phosphorylation at Raf-1 S43 and S259 by PKA only weakly inhibited EGF activation of Raf-1 and ERK when cells maintained high Raf-1 S338 phosphorylation. | | | 15899852
 |
Raf-1 kinase inhibitor protein: structure, function, regulation of cell signaling, and pivotal role in apoptosis.
Odabaei, Golaun, et al.
Adv. Cancer Res., 91: 169-200 (2004)
2004
显示摘要
The acquisition of resistance to conventional therapies such as radiation and chemotherapeutic drugs remains the major obstacle in the successful treatment of cancer patients. Tumor cells acquire resistance to apoptotic stimuli and it has been demonstrated that conventional therapies exert their cytotoxic activities primarily by inducing apoptosis in the cells. Resistance to radiation and chemotherapeutic drugs has led to the development of immunotherapy and gene therapy approaches with the intent of overcoming resistance to drugs and radiation as well as enhancing the specificity to eliminate tumor cells. However, cytotoxic lymphocytes primarily kill by apoptosis and, therefore, drug-resistant tumor cells may also be cross-resistant to immunotherapy. To evade apoptosis, tumor cells have adopted various mechanisms that interfere with the apoptotic signaling pathways and promote constitutive activation of cellular proliferation and survival pathways. Thus, modifications of the antiapoptotic genes in cancer cells are warranted for the effectiveness of conventional therapies as well as novel immunotherapeutic approaches. Such modifications will avert the resistant phenotype of the tumor cells and will render them susceptible to apoptosis. Current studies, both in vitro and preclinically in vivo, have been aimed at the modification and regulation of expression of apoptosis-related gene products and their activities. A novel protein designated Raf-1 kinase inhibitor protein (RKIP) has been partially characterized. RKIP is a member of the phosphatidylethanolamine-binding protein family. RKIP has been shown to disrupt the Raf-1-MEK1/2 [mitogen-activated protein kinase-ERK (extracellular signal-regulated kinase) kinase-1/2]-ERK1/2 and NF-kappaB signaling pathways, via physical interaction with Raf-1-MEK1/2 and NF-kappaB-inducing kinase or transforming growth factor beta-activated kinase-1, respectively, thereby abrogating the survival and antiapoptotic properties of these signaling pathways. In addition, RKIP has been shown to act as a signal modifier that enhances receptor signaling by inhibiting G protein-coupled receptor kinase-2. By regulating cell signaling, growth, and survival through its expression and activity, RKIP is considered to play a pivotal role in cancer, regulating apoptosis induced by drugs or immune-mediated stimuli. Overexpression of RKIP sensitizes tumor cells to chemotherapeutic drug-induced apoptosis. Also, induction of RKIP by drugs or anti-receptor antibodies sensitizes cancer cells to drug-induced apoptosis. In this review, we discuss the discovery, structure, function, and significance of RKIP in cancer. | | | 15327891
 |
Untying the regulation of the Raf-1 kinase.
Dhillon, Amardeep S and Kolch, Walter
Arch. Biochem. Biophys., 404: 3-9 (2002)
2002
显示摘要
The Raf-1 kinase is the entry point to the mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK-1/2) signaling pathway, which controls fundamental cellular functions including proliferation, differentiation, and survival. As such, Raf-1 is regulated by complex mechanisms that are incompletely understood. Recent results have shown that release from repression is an important event that facilitates the interaction of Raf-1 with the Ras activator and its substrate, MAPK/ERK-1/2 kinase. A number of distinct activation steps contribute in a combinatorial fashion to regulate and adjust Raf-1 activity. The efficiency of downstream signal transmission is modulated by protein:protein interactions, and new data consolidate an important role for kinase suppressor of ras (KSR) as a scaffolding protein. KSR is a dynamic scaffold whose function and localization is regulated by phosphorylation. | | | 12127063
 |
Anti-apoptotic signaling of the insulin-like growth factor-I receptor through mitochondrial translocation of c-Raf and Nedd4
Peruzzi, F., et al
J Biol Chem, 276:25990-6 (2001)
2001
| Immunoblotting (Western) | | 11352919
 |