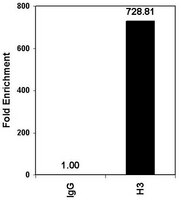
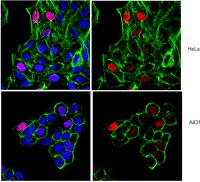
Total levels of hippocampal histone acetylation predict normal variability in mouse behavior.
Nesbitt, AM; McCurdy, RD; Bryant, SM; Alter, MD
PloS one
9
e94224
2014
Mostra il sommario
Genetic, pharmacological, and environmental interventions that alter total levels of histone acetylation in specific brain regions can modulate behaviors and treatment responses. Efforts have been made to identify specific genes that are affected by alterations in total histone acetylation and to propose that such gene specific modulation could explain the effects of total histone acetylation levels on behavior - the implication being that under naturalistic conditions variability in histone acetylation occurs primarily around the promoters of specific genes.Here we challenge this hypothesis by demonstrating with a novel flow cytometry based technique that normal variability in open field exploration, a hippocampus-related behavior, was associated with total levels of histone acetylation in the hippocampus but not in other brain regions.Results suggest that modulation of total levels of histone acetylation may play a role in regulating biological processes. We speculate in the discussion that endogenous regulation of total levels of histone acetylation may be a mechanism through which organisms regulate cellular plasticity. Flow cytometry provides a useful approach to measure total levels of histone acetylation at the single cell level. Relating such information to behavioral measures and treatment responses could inform drug delivery strategies to target histone deacetylase inhibitors and other chromatin modulators to places where they may be of benefit while avoiding areas where correction is not needed and could be harmful. | Fluorescence Activated Cell Sorting (FACS) | Mouse | 24788142
 |
Characterization of Toxoplasma gondii subtelomeric-like regions: identification of a long-range compositional bias that is also associated with gene-poor regions.
Dalmasso, MC; Carmona, SJ; Angel, SO; Agüero, F
BMC genomics
15
21
2014
Mostra il sommario
Chromosome ends are composed of telomeric repeats and subtelomeric regions, which are patchworks of genes interspersed with repeated elements. Although chromosome ends display similar arrangements in different species, their sequences are highly divergent. In addition, these regions display a particular nucleosomal composition and bind specific factors, therefore producing a special kind of heterochromatin. Using data from currently available draft genomes we have characterized these putative Telomeric Associated Sequences in Toxoplasma gondii.An all-vs-all pairwise comparison of T. gondii assembled chromosomes revealed the presence of conserved regions of ∼ 30 Kb located near the ends of 9 of the 14 chromosomes of the genome of the ME49 strain. Sequence similarity among these regions is ∼ 70%, and they are also highly conserved in the GT1 and VEG strains. However, they are unique to Toxoplasma with no detectable similarity in other Apicomplexan parasites. The internal structure of these sequences consists of 3 repetitive regions separated by high-complexity sequences without annotated genes, except for a gene from the Toxoplasma Specific Family. ChIP-qPCR experiments showed that nucleosomes associated to these sequences are enriched in histone H4 monomethylated at K20 (H4K20me1), and the histone variant H2A.X, suggesting that they are silenced sequences (heterochromatin). A detailed characterization of the base composition of these sequences, led us to identify a strong long-range compositional bias, which was similar to that observed in other genomic silenced fragments such as those containing centromeric sequences, and was negatively correlated to gene density.We identified and characterized a region present in most Toxoplasma assembled chromosomes. Based on their location, sequence features, and nucleosomal markers we propose that these might be part of subtelomeric regions of T. gondii. The identified regions display a unique trinucleotide compositional bias, which is shared (despite the lack of any detectable sequence similarity) with other silenced sequences, such as those making up the chromosome centromeres. We also identified other genomic regions with this compositional bias (but no detectable sequence similarity) that might be functionally similar. | | | 24417889
 |
Runx1t1 (Runt-related transcription factor 1; translocated to, 1) epigenetically regulates the proliferation and nitric oxide production of microglia.
Baby, N; Li, Y; Ling, EA; Lu, J; Dheen, ST
PloS one
9
e89326
2014
Mostra il sommario
Microglia, the resident immune cells of the brain, undergo rapid proliferation and produce several proinflammatory molecules and nitric oxide (NO) when activated in neuropathological conditions. Runx1t1 (Runt-related transcription factor 1, translocated to 1) has been implicated in recruiting histone deacetylases (HDACs) for transcriptional repression, thereby regulating cell proliferation. In the present study, Runx1t1 expression was shown to localize in amoeboid microglial cells of the postnatal rat brain, being hardly detectable in ramified microglia of the adult brain. Moreover, a marked expression of Runx1t1was induced and translocated to nuclei in activated microglia in vitro and in vivo. In view of these findings, it was hypothesized that Runx1t1 regulates microglial functions during development and in neuropathological conditions.siRNA-mediated knockdown of Runx1t1 significantly decreased the expression level of cell cycle-related gene, cyclin-dependent kinase 4 (Cdk4) and proliferation index in activated BV2 microglia. It was also shown that HDAC inhibitor (HDACi) treatment mimics the effects of Runx1t1 knockdown on microglial proliferation, confirming that microglial proliferation is associated with Runx1t1 expression and HDACs activity. Further, Runx1t1 and HDACs were shown to promote neurotoxic effect of microglia by repressing expression of LAT2, L-aminoacid transporter-2 (cationic amino acid transporter, y+ system), which normally inhibits NO production. This was confirmed by chromatin immunoprecipitation (ChIP) assay, which revealed that Runx1t1 binds to the promoter region of LAT2 and this binding increased upon microglial activation. However, the enhanced binding of Runx1t1 to the LAT2 promoter could not repress the LAT2 expression when the BV2 microglia cells were treated with HDACi, indicating that Runx1t1 requires HDACs to transcriptionally repress the expression of LAT2.In conclusion, it is suggested that Runx1t1 controls proliferation and the neurotoxic effect of microglia by epigenetically regulating Cdk4 and LAT2 via its interaction with HDACs. | | | 24586690
 |
PIAS1 regulates breast tumorigenesis through selective epigenetic gene silencing.
Liu, B; Tahk, S; Yee, KM; Yang, R; Yang, Y; Mackie, R; Hsu, C; Chernishof, V; O'Brien, N; Jin, Y; Fan, G; Lane, TF; Rao, J; Slamon, D; Shuai, K
PloS one
9
e89464
2014
Mostra il sommario
Epigenetic gene silencing by histone modifications and DNA methylation is essential for cancer development. The molecular mechanism that promotes selective epigenetic changes during tumorigenesis is not understood. We report here that the PIAS1 SUMO ligase is involved in the progression of breast tumorigenesis. Elevated PIAS1 expression was observed in breast tumor samples. PIAS1 knockdown in breast cancer cells reduced the subpopulation of tumor-initiating cells, and inhibited breast tumor growth in vivo. PIAS1 acts by delineating histone modifications and DNA methylation to silence the expression of a subset of clinically relevant genes, including breast cancer DNA methylation signature genes such as cyclin D2 and estrogen receptor, and breast tumor suppressor WNT5A. Our studies identify a novel epigenetic mechanism that regulates breast tumorigenesis through selective gene silencing. | | | 24586797
 |
Histone deacetylase expression in white matter oligodendrocytes after stroke.
Kassis, H; Chopp, M; Liu, XS; Shehadah, A; Roberts, C; Zhang, ZG
Neurochemistry international
77
17-23
2014
Mostra il sommario
Histone deacetylases (HDACs) constitute a super-family of enzymes grouped into four major classes (Class I-IV) that deacetylate histone tails leading to chromatin condensation and gene repression. Whether stroke-induced oligodendrogenesis is related to the expression of individual HDACs in the oligodendrocyte lineage has not been investigated. We found that 2 days after stroke, oligodendrocyte progenitor cells (OPCs) and mature oligodendrocytes (OLGs) were substantially reduced in the peri-infarct corpus callosum, whereas at 7 days after stroke, a robust increase in OPCs and OLGs was observed. Ischemic brains isolated from rats sacrificed 7 days after stroke were used to test levels of individual members of Class I (1 and 2) and Class II (4 and 5) HDACs in white matter oligodendrocytes during stroke-induced oligodendrogenesis. Double immunohistochemistry analysis revealed that stroke substantially increased the number of NG2+OPCs with nuclear HDAC1 and HDAC2 immunoreactivity and cytoplasmic HDAC4 which were associated with augmentation of proliferating OPCs, as determined by BrdU and Ki67 double reactive cells after stroke. A decrease in HDAC1 and an increase in HDAC2 immunoreactivity were detected in mature adenomatous polyposis coli (APC) positive OLGs, which paralleled an increase in newly generated BrdU positive OLGs in the peri-infarct corpus callosum. Concurrently, stroke substantially decreased the acetylation levels of histones H3 and H4 in both OPCs and OLGs. Taken together, these findings demonstrate that stroke induces distinct profiles of Class I and Class II HDACs in white matter OPCs and OLGs, suggesting that the individual members of Class I and II HDACs play divergent roles in the regulation of OPC proliferation and differentiation during brain repair after stroke. | | | 24657831
 |
Determinants of G quadruplex-induced epigenetic instability in REV1-deficient cells.
Schiavone, D; Guilbaud, G; Murat, P; Papadopoulou, C; Sarkies, P; Prioleau, MN; Balasubramanian, S; Sale, JE
The EMBO journal
33
2507-20
2014
Mostra il sommario
REV1-deficient chicken DT40 cells are compromised in replicating G quadruplex (G4)-forming DNA. This results in localised, stochastic loss of parental chromatin marks and changes in gene expression. We previously proposed that this epigenetic instability arises from G4-induced replication fork stalls disrupting the accurate propagation of chromatin structure through replication. Here, we test this model by showing that a single G4 motif is responsible for the epigenetic instability of the BU-1 locus in REV1-deficient cells, despite its location 3.5 kb from the transcription start site (TSS). The effect of the G4 is dependent on it residing on the leading strand template, but is independent of its in vitro thermal stability. Moving the motif to more than 4 kb from the TSS stabilises expression of the gene. However, loss of histone modifications (H3K4me3 and H3K9/14ac) around the transcription start site correlates with the position of the G4 motif, expression being lost only when the promoter is affected. This supports the idea that processive replication is required to maintain the histone modification pattern and full transcription of this model locus. | | | 25190518
 |
Plasmodium infection reduces the volume of the viral reservoir in SIV-infected rhesus macaques receiving antiretroviral therapy.
Zhan, XY; Wang, N; Liu, G; Qin, L; Xu, W; Zhao, S; Qin, L; Chen, X
Retrovirology
11
112
2014
Mostra il sommario
Previous studies indicated that Plasmodium infection activates the immune system, including memory CD4+ T cells, which constitute the reservoir of human immunodeficiency virus type-1 (HIV-1). Therefore, we postulated that co-infection with malaria might activate the reservoir of HIV-1. To test this hypothesis, we used a rhesus macaque model of co-infection with malaria and simian immunodeficiency virus (SIV), along with antiretroviral therapy (ART).Our results showed that Plasmodium infection reduced both the replication-competent virus pool in resting CD4+ T cells and the integrated virus DNA (iDNA) load in peripheral blood mononuclear cells in the monkeys. This reduction might be attributable to malaria-mediated activation and apoptotic induction of memory CD4+ T cells. Further studies indicated that histone acetylation and NF-kappaB (NF-κB) activation in resting CD4+ T cells may also play an important role in this reduction.The findings of this work expand our knowledge of the interaction between these two diseases. As more HIV-1-infected individuals in malaria-endemic areas receive ART, we should explore whether any of the patients co-infected with Plasmodium experience virologic benefits. | Immunoprecipitation | | 25487036
 |
Identification of STAT2 serine 287 as a novel regulatory phosphorylation site in type I interferon-induced cellular responses.
Steen, HC; Nogusa, S; Thapa, RJ; Basagoudanavar, SH; Gill, AL; Merali, S; Barrero, CA; Balachandran, S; Gamero, AM
The Journal of biological chemistry
288
747-58
2013
Mostra il sommario
STAT2 is a positive modulator of the transcriptional response to type I interferons (IFNs). STAT2 acquires transcriptional function by becoming tyrosine phosphorylated and imported to the nucleus following type I IFN receptor activation. Although most STAT proteins become dually phosphorylated on specific tyrosine and serine residues to acquire full transcriptional activity, no serine phosphorylation site in STAT2 has been reported. To find novel phosphorylation sites, mass spectrometry of immunoprecipitated STAT2 was used to identify several phosphorylated residues. Of these, substitution of serine 287 with alanine (S287A) generated a gain-of-function mutant that enhanced the biological effects of IFN-α. S287A-STAT2 increased cell growth inhibition, prolonged protection against vesicular stomatitis virus infection and enhanced transcriptional responses following exposure of cells to IFN-α. In contrast, a phosphomimetic STAT2 mutant (S287D) produced a loss-of-function protein that weakly activated IFN-induced ISGs. Our mechanistic studies suggest that S287A-STAT2 likely mediates its gain-of-function effects by prolonging STAT2/STAT1 dimer activation and retaining it in transcriptionally active complexes with chromatin. Altogether, we have uncovered that in response to type I IFN, STAT2 is serine phosphorylated in the coiled-coil domain that when phosphorylated can negatively regulate the biological activities of type I IFNs. | | | 23139419
 |
Lysine acetyltransferase GCN5 potentiates the growth of non-small cell lung cancer via promotion of E2F1, cyclin D1, and cyclin E1 expression.
Chen, L; Wei, T; Si, X; Wang, Q; Li, Y; Leng, Y; Deng, A; Chen, J; Wang, G; Zhu, S; Kang, J
J Biol Chem
288
14510-21
2013
Mostra il sommario
The lysine acetyltransferases play crucial but complex roles in cancer development. GCN5 is a lysine acetyltransferase that generally regulates gene expression, but its role in cancer development remains largely unknown. In this study, we report that GCN5 is highly expressed in non-small cell lung cancer tissues and that its expression correlates with tumor size. We found that the expression of GCN5 promotes cell growth and the G1/S phase transition in multiple lung cancer cell lines. Further study revealed that GCN5 regulates the expression of E2F1, cyclin D1, and cyclin E1. Our reporter assays indicated that the expression of GCN5 enhances the activities of the E2F1, cyclin D1, and cyclin E1 promoters. ChIP experiments suggested that GCN5 binds directly to these promoters and increases the extent of histone acetylation within these regions. Mechanistic studies suggested that GCN5 interacts with E2F1 and is recruited by E2F1 to the E2F1, cyclin D1, and cyclin E1 promoters. The function of GCN5 in lung cancer cells is abrogated by the knockdown of E2F1. Finally, we confirmed that GCN5 regulates the expression of E2F1, cyclin D1, and cyclin E1 and potentiates lung cancer cell growth in a mouse tumor model. Taken together, our results demonstrate that GCN5 specifically potentiates lung cancer growth by directly promoting the expression of E2F1, cyclin D1, and cyclin E1 in an E2F1-dependent manner. Our study identifies a specific and novel function of GCN5 in lung cancer development and suggests that the GCN5-E2F1 interaction represents a potential target for lung cancer treatment. | | | 23543735
 |
Histone modifications are responsible for decreased Fas expression and apoptosis resistance in fibrotic lung fibroblasts.
Huang, SK; Scruggs, AM; Donaghy, J; Horowitz, JC; Zaslona, Z; Przybranowski, S; White, ES; Peters-Golden, M
Cell death & disease
4
e621
2013
Mostra il sommario
Although the recruitment of fibroblasts to areas of injury is critical for wound healing, their subsequent apoptosis is necessary in order to prevent excessive scarring. Fibroproliferative diseases, such as pulmonary fibrosis, are often characterized by fibroblast resistance to apoptosis, but the mechanism(s) for this resistance remains elusive. Here, we employed a murine model of pulmonary fibrosis and cells from patients with idiopathic pulmonary fibrosis (IPF) to explore epigenetic mechanisms that may be responsible for the decreased expression of Fas, a cell surface death receptor whose expression has been observed to be decreased in pulmonary fibrosis. Murine pulmonary fibrosis was elicited by intratracheal injection of bleomycin. Fibroblasts cultured from bleomycin-treated mice exhibited decreased Fas expression and resistance to Fas-mediated apoptosis compared with cells from saline-treated control mice. Although there were no differences in DNA methylation, the Fas promoter in fibroblasts from bleomycin-treated mice exhibited decreased histone acetylation and increased histone 3 lysine 9 trimethylation (H3K9Me3). This was associated with increased histone deacetylase (HDAC)-2 and HDAC4 expression. Treatment with HDAC inhibitors increased Fas expression and restored susceptibility to Fas-mediated apoptosis. Fibroblasts from patients with IPF likewise exhibited decreased histone acetylation and increased H3K9Me3 at the Fas promoter and increased their expression of Fas in the presence of an HDAC inhibitor. These findings demonstrate the critical role of histone modifications in the development of fibroblast resistance to apoptosis in both a murine model and in patients with pulmonary fibrosis and suggest novel approaches to therapy for progressive fibroproliferative disorders. | Western Blotting | | 23640463
 |