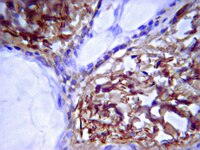
Mesenchymal stem cells in rabbit meniscus and bone marrow exhibit a similar feature but a heterogeneous multi-differentiation potential: superiority of meniscus as a cell source for meniscus repair.
Ding, Z; Huang, H
BMC musculoskeletal disorders
16
65
2015
Mostrar Resumo
The restoration of damaged meniscus has always been a challenge due to its limited healing capacity. Recently, bone marrow-derived mesenchymal stem cells (BMSCs) provide a promising alternative to repair meniscal defects. However, BMSCs are not ideal chondroprogenitor cells for meniscus repair because they have a high propensity for cartilage hypertrophy and bone formation. Our hypothesis is that mesenchymal stem cells (MSCs) reside in meniscus maintain specific traits distinct from others which may be more conducive to meniscus regeneration.MSCs were isolated from bone marrow and menisci of the rabbits. The similarities and differences between BMSCs and MMSCs were investigated in vitro by a cell culture model, ex vivo by a rabbit meniscus defect model and in vivo by a nude rat implantation model using histochemistry, immunocytochemistry, qRT-PCR and western blotting.Our data showed that two types of MSCs have universal stem cell characteristics including clonogenicity, multi-potency and self-renewal capacity. They both express stem cell markers including SSEA-4, Nanog, nucleostemin, strol-1, CD44 and CD90. However, MMSCs differed from BMSCs. MMSC colonies were much smaller and grew more slowly than BMSC colonies. Moreover, fewer MMSCs expressed CD34 than BMSCs. Finally, MMSCs always appeared a pronounced tendency to chondrogenic differentiation while BMSCs exhibited significantly greater osteogenic potential, whatever in vitro and in vivo.This study shows the similarities and differences between MMSCs and BMSCs for the first time. MMSCs are a promising source of mesenchymal stem cells in repairing meniscus defect. | 25887689
 |
Human articular chondrocytes express multiple gap junction proteins: differential expression of connexins in normal and osteoarthritic cartilage.
Mayan, MD; Carpintero-Fernandez, P; Gago-Fuentes, R; Martinez-de-Ilarduya, O; Wang, HZ; Valiunas, V; Brink, P; Blanco, FJ
The American journal of pathology
182
1337-46
2013
Mostrar Resumo
Osteoarthritis (OA) is the most common joint disease and involves progressive degeneration of articular cartilage. The aim of this study was to investigate if chondrocytes from human articular cartilage express gap junction proteins called connexins (Cxs). We show that human chondrocytes in tissue express Cx43, Cx45, Cx32, and Cx46. We also find that primary chondrocytes from adults retain the capacity to form functional voltage-dependent gap junctions. Immunohistochemistry experiments in cartilage from OA patients revealed significantly elevated levels of Cx43 and Cx45 in the superficial zone and down through the next approximately 1000 μm of tissue. These zones corresponded with regions damaged in OA that also had high levels of proliferative cell nuclear antigen. An increased number of Cxs may help explain the increased proliferation of cells in clusters that finally lead to tissue homeostasis loss. Conversely, high levels of Cxs in OA cartilage reflect the increased number of adjacent cells in clusters that are able to interact directly by gap junctions as compared with hemichannels on single cells in normal cartilage. Our data provide strong evidence that OA patients have a loss of the usual ordered distribution of Cxs in the damaged zones and that the reductions in Cx43 levels are accompanied by the loss of correct Cx localization in the nondamaged areas. | 23416160
 |
Human tendon stem cells better maintain their stemness in hypoxic culture conditions.
Zhang, J; Wang, JH
PloS one
8
e61424
2013
Mostrar Resumo
Tissues and organs in vivo are under a hypoxic condition; that is, the oxygen tension is typically much lower than in ambient air. However, the effects of such a hypoxic condition on tendon stem cells, a recently identified tendon cell, remain incompletely defined. In cell culture experiments, we subjected human tendon stem cells (hTSCs) to a hypoxic condition with 5% O2, while subjecting control cells to a normaxic condition with 20% O2. We found that hTSCs at 5% O2 had significantly greater cell proliferation than those at 20% O2. Moreover, the expression of two stem cell marker genes, Nanog and Oct-4, was upregulated in the cells cultured in 5% O2. Finally, in cultures under 5% O2, more hTSCs expressed the stem cell markers nucleostemin, Oct-4, Nanog and SSEA-4. In an in vivo experiment, we found that when both cell groups were implanted with tendon-derived matrix, more tendon-like structures formed in the 5% O2 treated hTSCs than in 20% O2 treated hTSCs. Additionally, when both cell groups were implanted with Matrigel, the 5% O2 treated hTSCs showed more extensive formation of fatty, cartilage-like and bone-like tissues than the 20% O2 treated cells. Together, the findings of this study show that oxygen tension is a niche factor that regulates the stemness of hTSCs, and that less oxygen is better for maintaining hTSCs in culture and expanding them for cell therapy of tendon injuries. | 23613849
 |
Isolation and characterization of novel, highly proliferative human CD34/CD73-double-positive testis-derived stem cells for cell therapy.
Choi, WY; Jeon, HG; Chung, Y; Lim, JJ; Shin, DH; Kim, JM; Ki, BS; Song, SH; Choi, SJ; Park, KH; Shim, SH; Moon, J; Jung, SJ; Kang, HM; Park, S; Chung, HM; Ko, JJ; Cha, KY; Yoon, TK; Kim, H; Lee, DR
Stem cells and development
22
2158-73
2013
Mostrar Resumo
Human adult stem cells are a readily available multipotent cell source that can be used in regenerative medicine. Despite many advantages, including low tumorigenicity, their rapid senescence and limited plasticity have curtailed their use in cell-based therapies. In this study, we isolated CD34/CD73-double-positive (CD34(+)/CD73(+)) testicular stromal cells (HTSCs) and found that the expression of CD34 was closely related to the cells' stemness and proliferation. The CD34(+)/CD73(+) cells grew in vitro for an extended period of time, yielding a multitude of cells (5.6×10(16) cells) without forming tumors in vivo. They also differentiated into all three germ layer lineages both in vitro and in vivo, produced cartilage more efficiently compared to bone marrow stem cells and, importantly, restored erectile function in a cavernous nerve crush injury rat model. Thus, these HTSCs may represent a promising new autologous cell source for clinical use. | 23509942
 |
Alternative splicing targeting the hTAF4-TAFH domain of TAF4 represses proliferation and accelerates chondrogenic differentiation of human mesenchymal stem cells.
Kazantseva, J; Kivil, A; Tints, K; Kazantseva, A; Neuman, T; Palm, K
PloS one
8
e74799
2013
Mostrar Resumo
Transcription factor IID (TFIID) activity can be regulated by cellular signals to specifically alter transcription of particular subsets of genes. Alternative splicing of TFIID subunits is often the result of external stimulation of upstream signaling pathways. We studied tissue distribution and cellular expression of different splice variants of TFIID subunit TAF4 mRNA and biochemical properties of its isoforms in human mesenchymal stem cells (hMSCs) to reveal the role of different isoforms of TAF4 in the regulation of proliferation and differentiation. Expression of TAF4 transcripts with exons VI or VII deleted, which results in a structurally modified hTAF4-TAFH domain, increases during early differentiation of hMSCs into osteoblasts, adipocytes and chondrocytes. Functional analysis data reveals that TAF4 isoforms with the deleted hTAF4-TAFH domain repress proliferation of hMSCs and preferentially promote chondrogenic differentiation at the expense of other developmental pathways. This study also provides initial data showing possible cross-talks between TAF4 and TP53 activity and switching between canonical and non-canonical WNT signaling in the processes of proliferation and differentiation of hMSCs. We propose that TAF4 isoforms generated by the alternative splicing participate in the conversion of the cellular transcriptional programs from the maintenance of stem cell state to differentiation, particularly differentiation along the chondrogenic pathway. | 24098348
 |
Bilayered constructs aimed at osteochondral strategies: the influence of medium supplements in the osteogenic and chondrogenic differentiation of amniotic fluid-derived stem cells.
Márcia T Rodrigues,Sang Jin Lee,Manuela E Gomes,Rui L Reis,Anthony Atala,James J Yoo,M Rodrigues
Acta biomaterialia
8
2012
Mostrar Resumo
The development of osteochondral tissue engineered interfaces would be a novel treatment for traumatic injuries and aging associated diseases that affect joints. This study reports the development of a bilayered scaffold, which consists of both bone and cartilage regions. On the other hand, amniotic fluid-derived stem cells (AFSCs) could be differentiated into either osteogenic or chondrogenic cells, respectively. In this study we have developed a bilayered scaffolding system, which includes a starch/polycaprolactone (SPCL) scaffold for osteogenesis and an agarose hydrogel for chondrogenesis. AFSC-seeded scaffolds were cultured for 1 or 2 weeks in an osteochondral-defined culture medium containing both osteogenic and chondrogenic differentiation factors. Additionally, the effect of the presence or absence of insulin-like growth factor-1 (IGF-1) in the culture medium was assessed. Cell viability and phenotypic expression were assessed within the constructs in order to determine the influence of the osteochondral differentiation medium. The results indicated that, after osteogenic differentiation, AFSCs that had been seeded onto SPCL scaffolds did not require osteochondral medium to maintain their phenotype, and they produced a protein-rich, mineralized extracellular matrix (ECM) for up to 2 weeks. However, AFSCs differentiated into chondrocyte-like cells appeared to require osteochondral medium, but not IGF-1, to synthesize ECM proteins and maintain the chondrogenic phenotype. Thus, although IGF-1 was not essential for creating osteochondral constructs with AFSCs in this study, the osteochondral supplements used appear to be important to generate cartilage in long-term tissue engineering approaches for osteochondral interfaces. In addition, constructs generated from agarose-SPCL bilayered scaffolds containing pre-differentiated AFSCs may be useful for potential applications in regeneration strategies for damaged or diseased joints. | 22510402
 |
Epithelial cell rests of Malassez contain unique stem cell populations capable of undergoing epithelial-mesenchymal transition.
Xiong, J; Mrozik, K; Gronthos, S; Bartold, PM
Stem cells and development
21
2012-25
2012
Mostrar Resumo
The epithelial cell rests of Malassez (ERM) are odontogenic epithelial cells located within the periodontal ligament matrix. While their function is unknown, they may support tissue homeostasis and maintain periodontal ligament space or even contribute to periodontal regeneration. We investigated the notion that ERM contain a subpopulation of stem cells that could undergo epithelial-mesenchymal transition and differentiate into mesenchymal stem-like cells with multilineage potential. For this purpose, ERM collected from ovine incisors were subjected to different inductive conditions in vitro, previously developed for the characterization of bone marrow mesenchymal stromal/stem cells (BMSC). We found that ex vivo-expanded ERM expressed both epithelial (cytokeratin-8, E-cadherin, and epithelial membrane protein-1) and BMSC markers (CD44, CD29, and heat shock protein-90β). Integrin α6/CD49f could be used for the enrichment of clonogenic cell clusters [colony-forming units-epithelial cells (CFU-Epi)]. Integrin α6/CD49f-positive-selected epithelial cells demonstrated over 50- and 7-fold greater CFU-Epi than integrin α(6)/CD49f-negative cells and unfractionated cells, respectively. Importantly, ERM demonstrated stem cell-like properties in their differentiation capacity to form bone, fat, cartilage, and neural cells in vitro. When transplanted into immunocompromised mice, ERM generated bone, cementum-like and Sharpey's fiber-like structures. Additionally, gene expression studies showed that osteogenic induction of ERM triggered an epithelial-mesenchymal transition. In conclusion, ERM are unusual cells that display the morphological and phenotypic characteristics of ectoderm-derived epithelial cells; however, they also have the capacity to differentiate into a mesenchymal phenotype and thus represent a unique stem cell population within the periodontal ligament. | 22122577
 |
Advanced glycation end products cause collagen II reduction by activating Janus kinase/signal transducer and activator of transcription 3 pathway in porcine chondrocytes.
Huang, CY; Lai, KY; Hung, LF; Wu, WL; Liu, FC; Ho, LJ
Rheumatology (Oxford, England)
50
1379-89
2011
Mostrar Resumo
The major risk factor for OA is ageing; however, the mechanisms remain largely unclear. We investigated the effects and mechanisms of advanced glycation end products (AGEs) that accumulate in aged joints in chondrocytes.Porcine chondrocytes or cartilage fragments were prepared. Gene expression of MMPs and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) was assessed by real-time RT-PCR. Gelatin zymography was used to determine MMP-13 enzyme activity. Histochemistry or immunoblotting analysis was applied to determine the expression of collagen II, proteoglycan and aggrecan. Electrophoretic mobility shift assay and immunoblotting were used to study the activation of signal transducer and activator of transcription 3 (STAT3). Genetic manipulations with short hairpin RNA (shRNA) or dominant negative constructs were applied.AGE enhanced expression and enzyme activity of MMP and ADAMTS genes and resulted in reduction of collagen II. Both janus kinase 2 (JAK2) and JAK3 inhibitors suppressed AGE-induced MMP-13, ADAMTS-4 and ADAMTS-5 expression and enzyme activity. Inhibition of JAK2 or JAK3 prevented AGE-mediated decrease of collagen II in chondrocytes and proteoglycan (aggrecan) degradation in cartilage fragments. In addition, interference of STAT3 expression inhibited AGE-induced MMP-13 and ADAMTS enzyme activities and mRNA levels. Furthermore, expression of the dominant negative receptor of AGE (DN-RAGE) blocked AGE-induced STAT3 phosphorylation.Blocking JAK/STAT3 signalling pathway inhibited AGE-induced activation of MMP-13 and ADAMTS and prevented AGE-mediated decrease of collagen II and proteoglycan (aggrecan). The results indicated that JAK/STAT3 pathway may be a potential target for designing disease-modifying drugs for the treatment of OA. | 21482542
 |
Preservation of the chondrocyte's pericellular matrix improves cell-induced cartilage formation.
Lucienne A Vonk,Behrouz Zandieh Doulabi,Chunling Huang,Marco N Helder,Vincent Everts,Ruud A Bank
Journal of cellular biochemistry
110
2010
Mostrar Resumo
The extracellular matrix surrounding chondrocytes within a chondron is likely to affect the metabolic activity of these cells. In this study we investigated this by analyzing protein synthesis by intact chondrons obtained from different types of cartilage and compared this with chondrocytes. Chondrons and chondrocytes from goats from different cartilage sources (articular cartilage, nucleus pulposus, and annulus fibrosus) were cultured for 0, 7, 18, and 25 days in alginate beads. Real-time polymerase chain reaction analyses indicated that the gene expression of Col2a1 was consistently higher by the chondrons compared with the chondrocytes and the Col1a1 gene expression was consistently lower. Western blotting revealed that Type II collagen extracted from the chondrons was cross-linked. No Type I collagen could be extracted. The amount of proteoglycans was higher for the chondrons from articular cartilage and nucleus pulposus compared with the chondrocytes, but no differences were found between chondrons and chondrocytes from annulus fibrosus. The expression of both Mmp2 and Mmp9 was higher by the chondrocytes from articular cartilage and nucleus pulposus compared with the chondrons, whereas no differences were found with the annulus fibrosus cells. Gene expression of Mmp13 increased strongly by the chondrocytes (>>50-fold), but not by the chondrons. Taken together, our data suggest that preserving the pericellular matrix has a positive effect on cell-induced cartilage production. | 20213765
 |
Human mesenchymal stem cell culture for neural transplantation.
David Gordon, Neil J Scolding, David Gordon, Neil J Scolding, David Gordon, Neil J Scolding
Methods in molecular biology (Clifton, N.J.)
549
103-18
2009
Mostrar Resumo
Mesenchymal stem cells (MSCs) have the potential to play a role in autologous repair of central nervous system injury or disease, circumventing both the complications associated with immune rejection of allogenic cells, and many of the ethical concerns associated with embryonic stem cell use. Human bone marrow-derived MSCs can be extracted relatively simply from the marrow of adult patients and maintained and expanded in culture. More importantly, it has been previously demonstrated that MSCs have the capacity to differentiate into neurons and glia in vitro when grown under appropriate conditions. Multipotent MSCs have also been successfully used in transplantation studies in animal models of disease as diverse as demyelination, stroke, trauma and Parkinson's disease. MSCs therefore provide an attractive and practical source of stem cells for reparative therapy in patients, and in this paper we describe methods for the reproducible culture and neural differentiation of human MSCs generated from patient marrow. | 19378199
 |