Epstein-Barr virus-induced epigenetic alterations following transient infection.
Queen, KJ; Shi, M; Zhang, F; Cvek, U; Scott, RS
International journal of cancer. Journal international du cancer
132
2076-86
2013
Afficher le résumé
Epstein-Barr virus (EBV) is a known tumor virus associated with an increasing array of malignancies; however, the association of the virus with certain malignancies is often erratic. To determine EBV's contributions to tumorigenesis in a setting of incomplete association, a transient model of infection was established where a clonal CCL185 carcinoma cell line infected with recombinant EBV was allowed to lose viral genomes by withdrawal of selection pressure. Global gene expression comparing EBV-negative, transiently infected clones to uninfected controls identified expression changes in more than 1,000 genes. Among downregulated genes, several genes known to be deoxyribonucleic acid (DNA) methylated in cancer were identified including E-cadherin and PYCARD. A cadherin switch, increased motility and enhanced cellular invasiveness present in EBV-positive cells were retained after viral loss, indicating an epigenetic effect. Repression of PYCARD expression was a result of increased promoter CpG methylation, whereas loss of E-cadherin expression after transient EBV infection did not correlate with increased DNA methylation of the E-cadherin promoter. Rather, repression of E-cadherin was consistent with the formation of a repressive chromatin state. Decreased histone 3 or 4 acetylation at the promoter and 5' end of the E-cadherin gene was observed in an EBV-negative, transiently infected clone relative to the uninfected controls. These results suggest that EBV can stably alter gene expression in a heritable fashion in formerly infected cells, whereas its own contribution to the oncogenic process is masked. | Western Blotting | 23047626
 |
TMS1, a novel proapoptotic caspase recruitment domain protein, is a target of methylation-induced gene silencing in human breast cancers.
Conway, K E, et al.
Cancer Res., 60: 6236-42 (2000)
1999
Afficher le résumé
Gene silencing associated with aberrant methylation of promoter region CpG islands is an acquired epigenetic alteration that serves as an alternative to genetic defects in the inactivation of tumor suppressor and other genes in human cancers. The hypothesis that aberrant methylation plays a direct causal role in carcinogenesis hinges on the question of whether aberrant methylation is sufficient to drive gene silencing. To identify downstream targets of methylation-induced gene silencing, we used a human cell model in which aberrant CpG island methylation is induced by ectopic expression of DNA methyltransferase. Here we report the isolation and characterization of TMS1 (target of methylation-induced silencing), a novel CpG island-associated gene that becomes hypermethylated and silenced in cells overexpressing DNA cytosine-5-methyltransferase-1. We also show that TMS1 is aberrantly methylated and silenced in human breast cancer cells. Forty percent (11 of 27) of primary breast tumors exhibited aberrant methylation of TMS1. TMS1 is localized to chromosome 16p11.2-12.1 and encodes a 22-kDa predicted protein containing a COOH-terminal caspase recruitment domain, a recently described protein interaction motif found in apoptotic signaling molecules. Ectopic expression of TMS1 induced apoptosis in 293 cells and inhibited the survival of human breast cancer cells. The data suggest that methylation-mediated silencing of TMS1 confers a survival advantage by allowing cells to escape from apoptosis, supporting a new role for aberrant methylation in breast tumorigenesis. | | 11103776
 |
Activation of a caspase-9-mediated apoptotic pathway by subcellular redistribution of the novel caspase recruitment domain protein TMS1.
McConnell, B B and Vertino, P M
Cancer Res., 60: 6243-7 (2000)
1999
Afficher le résumé
Genetic and epigenetic alterations affecting proteins involved in apoptosis can contribute to the establishment and progression of cancer. Recently, our laboratory has isolated a novel gene, TMS1, that is aberrantly methylated and silenced in a significant proportion of human breast cancers. TMS1 contains a caspase recruitment domain (CARD), suggesting a role in caspase-mediated cell death. In the present study, we characterize the participation of TMS1 in apoptosis and examine the subcellular localization of the protein. Inducible expression of TMS1 inhibited cellular proliferation and induced DNA fragmentation in a time-dependent manner. These apoptotic events were blocked by the general caspase inhibitor, Z-VAD-fmk. The ability of TMS1 to trigger apoptosis was also suppressed by a dominant negative form of caspase-9 but not by a dominant negative form of caspase-8, indicating that TMS1 functions through activation of caspase-9. Unlike a number of other CARD-containing proteins, TMS1 did not activate nuclear factor kappaB-dependent transcription, consistent with a proapoptotic role for TMS1 in death signaling pathways. Timed localization studies revealed that TMS1-induced apoptosis was accompanied by the redistribution of TMS1 from the cytoplasm to perinuclear spherical structures. Whereas the apoptotic activity of TMS1 was blocked by caspase inhibition, the formation of TMS1-containing subcellular structures was not, suggesting that the redistribution of TMS1 precedes caspase activation. Both the proapoptotic activity of TMS1 and aggregate formation were dependent on the CARD. In summary, the data indicate that TMS1-induced apoptosis proceeds through a CARD-dependent aggregation step followed by activation of a caspase-9-mediated pathway. | | 11103777
 |
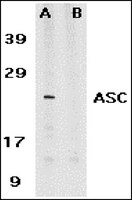
ASC, a novel 22-kDa protein, aggregates during apoptosis of human promyelocytic leukemia HL-60 cells.
Masumoto, J, et al.
J. Biol. Chem., 274: 33835-8 (1999)
1998
Afficher le résumé
The cytoskeletal and/or nuclear matrix molecules responsible for morphological changes associated with apoptosis were identified using monoclonal antibodies (mAbs). We developed mAbs against Triton X-100-insoluble components of HL-60 cells pretreated with all-trans retinoic acid. In particular, one mAb recognized a 22-kDa protein that exhibited intriguing behavior by forming an aggregate and appearing as a speck during apoptosis induced by retinoic acid and other anti-tumor drugs. Cloning and sequencing of its cDNA revealed that this protein comprises 195 amino acids and that its C-terminal half has a caspase recruitment domain (CARD) motif, characteristic of numerous proteins involved in apoptotic signaling. We referred to this protein as ASC (apoptosis-associated speck-like protein containing a CARD). The ASC gene was mapped on chromosome 16p11.2-12. The antisense oligonucleotides of ASC were found to reduce the expression of ASC, and consequently, etoposide-mediated apoptosis of HL-60 cells was suppressed. Our results indicate that ASC is a novel member of the CARD-containing adaptor protein family. | | 10567338
 |